Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
AdipoGen Life Sciences
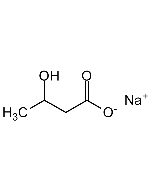
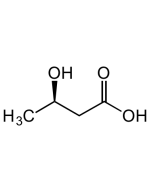
(S)-3-Hydroxybutyric acid
40
CHF
CHF 40.00
In stock
AG-CR1-3617-M02525 mgCHF 40.00
AG-CR1-3617-M100100 mgCHF 95.00

| Product Details | |
|---|---|
| Synonyms | (S)-β-Hydroxybutyric acid; L-(+)-3-Hydroxybutyric acid; L-(+)-BHB |
| Product Type | Chemical |
| Properties | |
| Formula |
C4H8O3 |
| MW | 104.1 |
| Merck Index | 14: 4816 |
| CAS | 6168-83-8 |
| Purity Chemicals | ≥98% (TLC) |
| Appearance | White solid. |
| Solubility | Soluble in water, 100% ethanol or methanol. |
| Identity | Determined by 1H-NMR. |
| Other Product Data |
Optical Rotation: [α]20/D c=6 in H2O: 25°±1.0° |
| InChi Key | WHBMMWSBFZVSSR-VKHMYHEASA-N |
| Smiles | C[C@H](O)CC(O)=O |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice | Keep cool and dry. |
| Use/Stability | Stable for at least 2 years after receipt when stored at -20°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
- Physiologically insignificant stereoisomer of (R)-3-Hydroxybutyric acid, a key metabolite of the ketolytic pathway.
- Anticonvulsant.
- Endogenous inhibitor of histone deacetylases (HDACs) 1, 3 and 4.
- Ligand of free fatty acid receptor 3 (FFAR3; GPR41) and hydroxycarboxylic acid receptor 2 (HCAR2; GPR109B).
- NLRP3 inflammasome inhibitor. Prevents K+-efflux and reduces ASC oligomerization and speck formation.
Product References
- Effects of beta-hydroxy butyric acid on insulin binding to its receptor and on autophosphorylation of the receptor: H. Ohtusaka, et al.; Endocrinol. Jpn. 37, 915 (1990)
- Acetoacetate, acetone, and dibenzylamine (a contaminant in L-(+)-beta-hydroxybutyrate) exhibit direct anticonvulsant actions in vivo: J.M. Rho, et al.; Epilepsia 43, 358 (2002)
- The direct measurement of 3-beta-hydroxy butyrate enhances the management of diabetic ketoacidosis in children and reduces time and costs of treatment: M. Vanelli, et al.; Diabetes Nutr. Metab. 16, 312 (2003)
- Detection of cerebral {beta}-hydroxy butyrate, acetoacetate, and lactate on proton MR spectroscopy in children with diabetic ketoacidosis: S.L. Wootton-Gorges, et al.; AJNR 26, 1286 (2005)
- β-Hydroxybutyrate activates the NF-κB signaling pathway to promote the expression of pro-inflammatory factors in calf hepatocytes: X. Shi, et al.; Cell. Physiol. Biochem. 33, 920 (2014)
- β-hydroxybutyrate: Much more than a metabolite: J. C. Newman & E. Verdin; Diab. Res. Clin. Pract. 106, 173 (2014) (Review)
- BHBA suppresses LPS-induced inflammation in BV-2 cells by inhibiting NF-κB activation: S.P. Fu, et al.; Med. Inflamm. 2014, ID983401 (2014)
- Anti-inflammatory effects of BHBA in both in vivo and in vitro Parkinson's disease models are mediated by GPR109A-dependent mechanisms: S.P. Fu, et al.; J. Neuroinflamm. 12, ID9 (2015)
- The ketone metabolite β-hydroxybutyrate blocks NLRP3 inflammasome-mediated inflammatory disease: Y.H. Youm, et al.; Nat. Med. 21, 263 (2015)
- Taming the inflammasome: M. Levy, et al.; Nat. Med. 21, 213 (2015)
- Inflammasome inhibition: putting out the fire: M.G. Netea & L.A. Joosten; Cell Metab. 21, 513 (2015)
- Inflammasome: starving inflammation: E. Kugleberg; Nat. Rev. Immunol. 15, 199 (2015) (Review)
- The Nlrp3 inflammasome admits defeat: C.J. Gross & O. Gross; Trends Immunol. 36, 323 (2015) (Review)