Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
AdipoGen Life Sciences
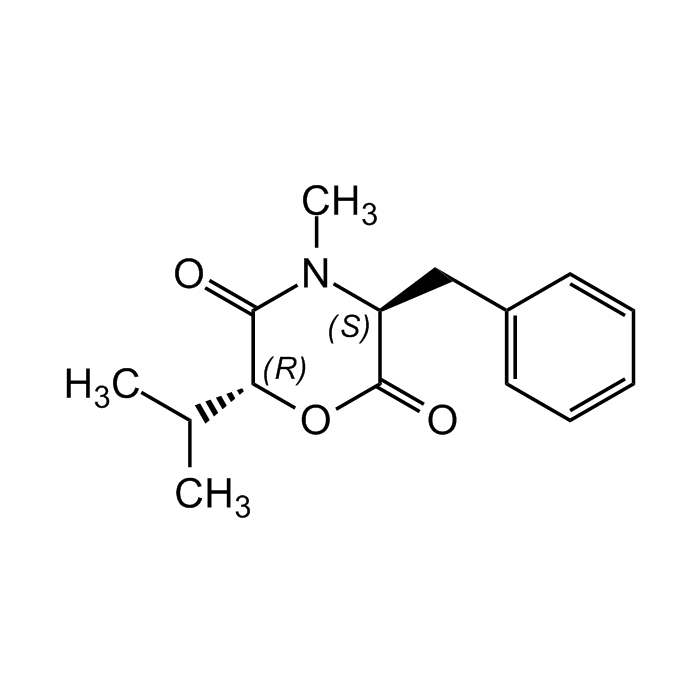
(3S,6R)-Lateritin
As low as
230
CHF
CHF 230.00
In stock
Only %1 left
AG-CN2-0042-M0011 mgCHF 230.00
| Product Details | |
|---|---|
| Synonyms | (3S,6R)-Bassiatin; Antibiotic PI290 |
| Product Type | Chemical |
| Properties | |
| Formula | C15H19NO3 |
| MW | 261.3 |
| CAS | 65454-13-9 |
| Source/Host Chemicals | Isolated from fungus Gibberella sp. |
| Purity Chemicals | ≥97% (HPLC) |
| Appearance | Off-white solid. |
| Solubility | Soluble in ethanol or DMSO. |
| Identity | Determined by 1H-NMR. |
| InChi Key | YOKBTBNVNCFOBF-QWHCGFSZSA-N |
| Smiles | CC(C)[C@H]1OC(=O)[C@H](CC2=CC=CC=C2)N(C)C1=O |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice |
Keep cool and dry. Protect from light. |
| Use/Stability |
Stable for at least 2 years after receipt when stored at -20°C. Store solutions at -20°C in the dark. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
- N-methylated peptide that is structurally equivalent to a monomer of beauvericin (Prod. No. AG-CN2-0043) [3, 4].
- Acyl-CoA:cholesterol acyltransferase (ACAT) inhibitor [1, 5].
- Platelet aggregation inhibitor [2, 5].
- Antimicrobial [5].
- Note: The (3R,6R)-stereoisomer was shown to have anti-cancer and apoptosis inducing activity [5-7].
Product References
- Lateritin, a new inhibitor of acyl-CoA:cholesterol acyltransferase produced by Gibberella lateritium IFO 7188: K. Hasumi, et al.; J. Antibiot. (Tokyo). 46, 1782 (1993)
- Bassiatin, a new platelet aggregation inhibitor produced by Beauveria bassiana K-717: T. Kagamizono, et al.; J. Antibiot. (Tokyo). 48, 1407 (1995)
- Beauvericin induces cytotoxic effects in human acute lymphoblastic leukemia cells through cytochrome c release, caspase 3 activation: the causative role of calcium: G.M. Jow, et al.; Cancer Lett. 216, 165 (2004)
- Beauvericin activates Ca2+-activated Cl- currents and induces cell deaths in Xenopus oocytes via influx of extracellular Ca2+: C.Y. Tang, et al.; Chem. Res. Toxicol. 18, 825 (2005)
- Isolation of human cancer cell growth inhibitory, antimicrobial lateritin from a mixed fungal culture: R.K. Pettit, et al.; Planta Med. 76, 500 (2010)
- (3R,6R)-4-methyl-6-(1-methylethyl)-3-phenylmethyl-perhydro-1,4-oxazine-2,5-dione: an apoptosis-inducer from the fruiting bodies of Isaria japonica: H. Oh, et al.; Planta Med. 68, 345 (2002)
- A novel antioestrogen agent (3R,6R)-bassiatin inhibits cell proliferation and cell cycle progression by repressing cyclin D1 expression in 17β-oestradiol-treated MCF-7 cells: L. Meng, et al.; Cell Biol. Int. 35, 599 (2011)