Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
Latrunculin A & B - Potent Actin Polymerization Inhibitors
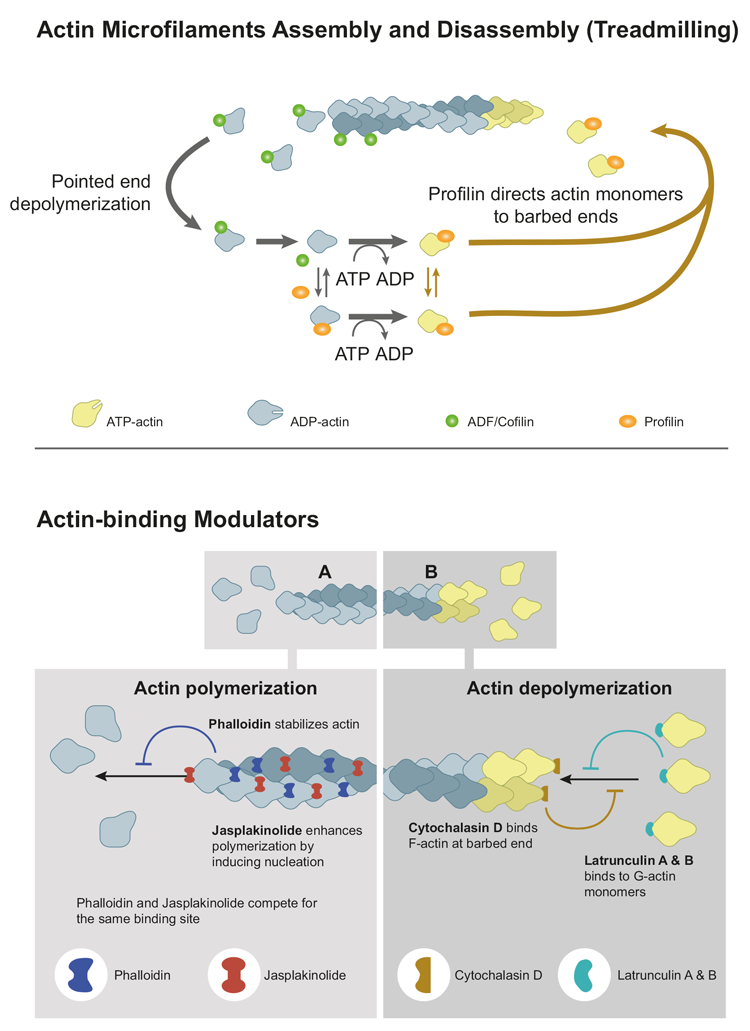
Microfilaments, also known as actin filaments, are one of the three main types of cytoskeletal filaments found within eukaryotic cells. They are composed of globular protein subunits called actin monomers that polymerize to form long, thin fibers. Actin must be bound to ATP to assemble into a polymer. The actin filament itself has structural polarity similar to microtubules. There are two distinct ends to the filament, called the "(-)" end and the "(+)" end. At the "(+)" end, actin subunits are added onto the elongating filament, and at the "(-)" end, actin subunits are disassembled. The ability of the actin filament to assemble and disassemble, thereby changing its length, is termed dynamic instability. This dynamic instability is integral to some cellular functions. The dynamic assembly and disassembly of microfilaments are regulated by a variety of proteins, including actin-binding proteins. These proteins control the polymerization and depolymerization of actin monomers, allowing cells to rapidly reorganize their cytoskeleton in response to different stimuli.
Microfilaments play a crucial role in i) the maintenance of the cell's shape and structural integrity; ii) in cell motility, which includes processes like cell crawling, amoeboid movement and muscle contraction; iii) in cytokinesis, the process that divides the cell's cytoplasm into two daughter cells; iv) in cell adhesion by interacting with integrin proteins in the cell membrane; v) in intracellular transport of organelles and vesicles; vi) in endocytosis and exocytosis, being involved in vesicle formation, movement, and fusion with the cell membrane; and vii) in signal transduction, transmitting mechanical signals within the cell, which can impact various cellular responses, such as gene expression and cell growth.
Figure: Schematic overview on actin microfilament assembly and disassembly, and on potential interactions of actin-binding small molecule modulators.
Latrunculin A & B
Latrunculins (including Latrunculin A & B) are natural compounds and a class of toxins originally isolated from the marine sponge Latrunculia magnifica. These compounds have been found to exhibit potent effects on actin polymerization, which is a crucial process for maintaining the cell's structural integrity, cell motility, cell division and other cellular functions. Latrunculins bind to actin monomers in a 1:1 ratio and prevent them from polymerizing into filaments. This leads to the disassembly of existing actin filaments and the inhibition of new filament formation. Similar to cytochalasins, latrunculins are well-known cell-disrupting agents.
Latrunculins are used as research tools in cell biology and molecular studies to investigate the roles of actin in various cellular processes. Additionally, these compounds have shown potential in cancer research due to their ability to inhibit cell migration and invasion, which are processes associated with the metastasis of cancer cells.
Literature References:
- Latrunculins: novel marine toxins that disrupt microfilament organization in cultured cells: I. Spector, et al.; Science 219, 493 (1983)
- Latrunculin A is a potent inhibitor of phagocytosis by macrophages: C.A. de Oliveira & B. Mantovani; Life Sci. 43, 1825 (1988)
- Use of latrunculin-A, an actin monomer-binding drug: K. Ayscough; Methods Enzymol. 298, 18 (1998)
- Actin-latrunculin A structure and function. Differential modulation of actin-binding protein function by latrunculin A: E.G. Yarmola, et al.; J. Biol. Chem. 275, 28120 (2000)
- Latrunculin A has a strong anticancer effect in a peritoneal dissemination model of human gastric cancer in mice: H. Konishi, et al.; Anticancer Res. 29, 2091 (2009)
- Inhibition of interdomain motion in g-actin by the natural product latrunculin: a molecular dynamics study: S. Rennebaum & A. Caflisch; Proteins 80, 1998 (2012)
- Latrunculin A - Insight into Specific Modifications to Design Novel Drugs to Overcome Resistance: R. Lalitha, et al.; Curr. Comput. Aided Drug Dev. 12, 107 (2016)
- Polymerization and depolymerization of actin with nucleotide states at filament ends: I. Fujiwara, et al.; Biophys. Rev. 10, 1513 (2018)
- Discovery of cytotoxic natural products from Red Sea sponges: Structure and synthesis: S. Khan, et al.; Eur. J. Med. Chem. 220, 113491 (2021)
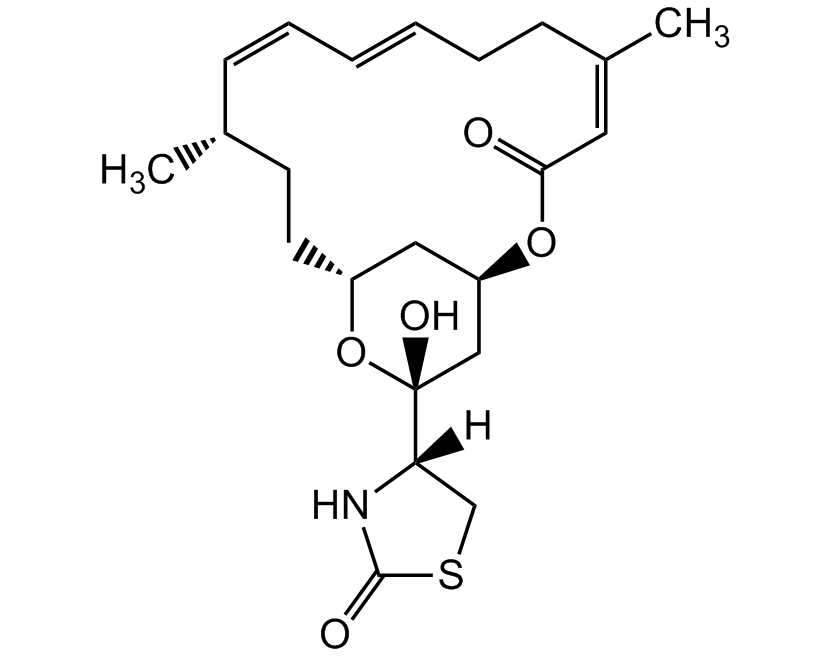
Latrunculin A
Latrunculin A is a cell permeable marine toxin, that disrupts microfilament-mediated processes. It is an actin polymerization inhibitor in vitro and in vivo by the formation of a 1:1 complex with monomeric G-actin and it depolymerizes actin filaments (F-actin). It is used as a potent phagocytosis inhibitor and anticancer compound, inhibiting tumor cell invasion.
AG-CN2-0027 (100 µg, 500 µg and BULK)
AdipoGen Life Sciences is an original Manufacturer of Latrunculin A. BULK quantities are available from Stock!
|
Product Specifications: CAS: 76343-93-6Source: Isolated from Latrunculia magnifica Purity: >97% HPLC Identity: Determined by 1H-NMR Contact us at info@adipogen.com and inquire about BULK Pricing! |
 |
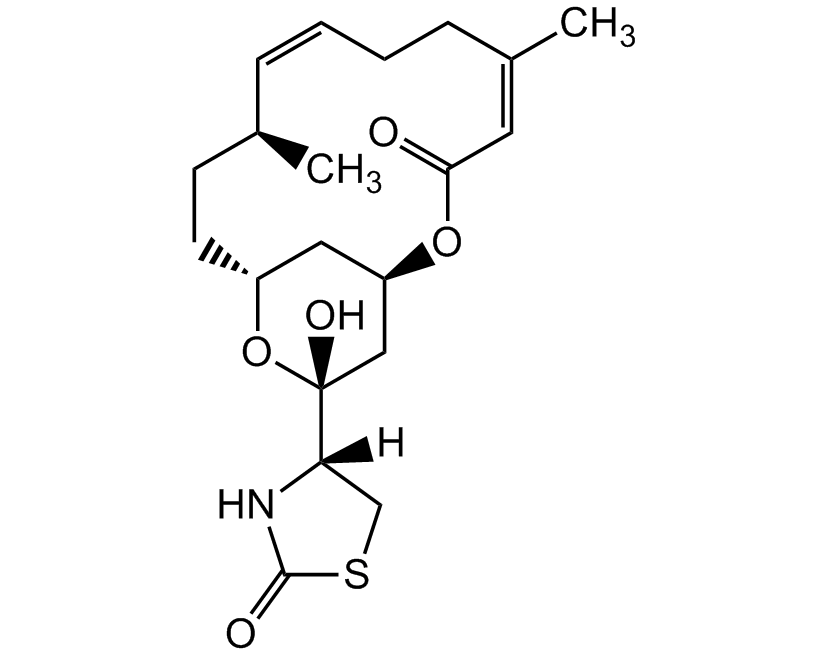
Latrunculin B
Latrunculin B is a cell permeable marine toxin, that disrupts microfilament-mediated processes. It is an actin polymerization inhibitor in vitro and in vivo by the formation of a 1:1 complex with monomeric G-actin and it depolymerizes actin filaments (F-actin). It is less potent than latrunculin A and may have fewer unwanted effects than latrunculin A.
AG-CN2-0031 (500 µg, 1 mg and BULK)
AdipoGen Life Sciences is the Key Manufacturer of Latrunculin B. BULK quantities are available from Stock!
|
Product Specifications: CAS: 76343-94-7Source: Isolated from Latrunculia magnifica Purity: >98% HPLC Identity: Determined by 1H-NMR Contact us at info@adipogen.com and inquire about BULK Pricing! |
 |
| Product Name | PID | Product Description |
| Cytochalasin B | AG-CN2-0504 | Actin polymerization inhibitor. Binds to the barbed end of actin, reversibly inhibiting the elongation and shortening of actin filaments. Induces nuclear extrusion. By disrupting actin polymerization, blocks diverse cellular functions, including cell division, migration, phagocytosis, exocytosis and chemotaxis. Induces DNA fragmentation. Used in cytoskeletal reorganization, cell imaging and organelle trafficking studies. |
| Cytochalasin H | BVT-0447 | Actin polymerization inhibitor. Used in actin polymerization and cytoskeletal reorganization studies. |
| Cytochalasin J | BVT-0450 | Actin polymerization inhibitor. Affects chromosome congression and spindle microtubule organization in PtK1 cells. |
| Jasplakinolide (high purity) | AG-CN2-0037 | Cell permeable, non-fluorescent F-actin probe. Potent inducer of actin polymerization and stabilization. A research reagent used for autophagy/phagocytosis research. |
| Latrunculin A | AG-CN2-0027 | Actin polymerization inhibitor in vitro and in vivo by the formation of a 1:1 complex with monomeric G-actin. Depolymerizes actin filaments (F-actin). Potent phagocytosis inhibitor. |
| Latrunculin B | AG-CN2-0031 | Actin polymerization inhibitor in vitro and in vivo by the formation of a 1:1 complex with monomeric G-actin. Depolymerizes actin filaments (F-actin). Less potent than Latrunculin A. |
| 16-epi-Latrunculin B | AG-CN2-0034 | Depolymerizes actin filaments (F-actin). |
| Sceptrin . dihydrochloride | AG-CN2-0440 | Actin polymerization inhibitor. Direct binding with monomeric actin disrupts the polymerization of actin into microfilaments necessary for cellular mechanical processes. |
