Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
AdipoGen Life Sciences
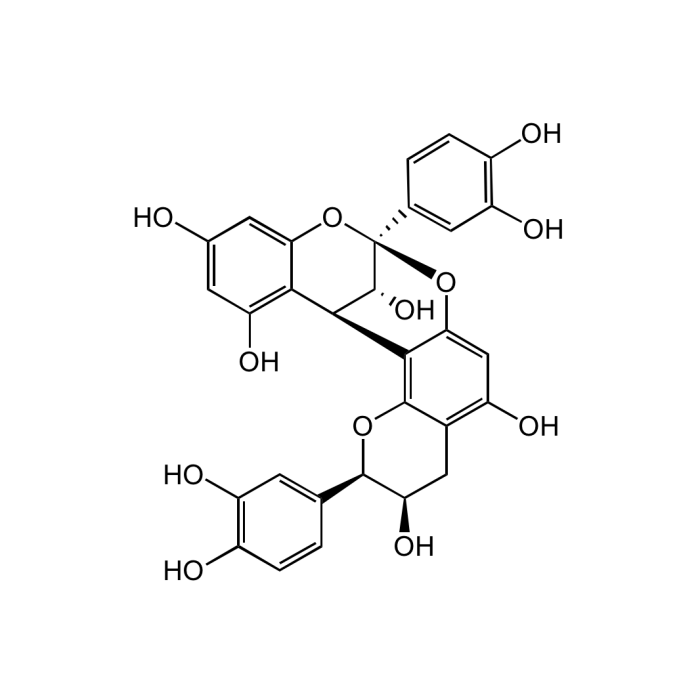
Procyanidin A2
As low as
60
CHF
CHF 60.00
In stock
Only %1 left
AG-CN2-0486-M0011 mgCHF 60.00
AG-CN2-0486-M0055 mgCHF 180.00
AG-CN2-0486-M02525 mgCHF 630.00
| Product Details | |
|---|---|
| Synonyms | (+)-Epicatechin-(4β-8,2β-O-7)-epicatechin; PCA2; Proanthocyanidin A2; PA2; Procyanidol A2; Procyanidin dimer A2 |
| Product Type | Chemical |
| Properties | |
| Formula |
C30H24O12 |
| MW | 576.5 |
| CAS | 41743-41-3 |
| Source/Host Chemicals | Isolated from grape seeds (Vitis vinifera L). |
| Purity Chemicals | ≥95% (NMR) |
| Appearance | Pale pink solid. |
| Solubility | Soluble in DMSO, 100% ethanol or methanol. Sparingly soluble in water (0.1 mg/ml). |
| Identity | Determined by 1H-NMR. |
| InChi Key | NSEWTSAADLNHNH-LSBOWGMISA-N |
| Smiles | OC1=CC(O)=C([C@H](C2=C(O[C@H](C3=CC(O)=C(O)C=C3)[C@H](O)C4)C4=C(O)C=C2O5)[C@@H](O)[C@@]5(C6=CC(O)=C(O)C=C6)O7)C7=C1 |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice |
Keep cool and dry. Protect from light. Protect from moisture and oxygen. |
| Use/Stability | Stable for at least 2 years after receipt when stored at -20°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
- A-Type proanthocyanidine.
- Potent antioxidant. Free radical scavenger and lipid peroxidation inhibitor.
- Potent low-density lipoprotein (LDL) oxidation inhibitor.
- Antidiabetic. Prevents islet cell apoptosis, hyperglycemia and type 2 diabetes through it's antioxidant properties.
- Antibacterial agent.
- Antiviral. Shown to inhibit viral replication of canine distemper virus (CDV).
- Glycosyltransferase B (GTFB) inhibitor.
Product References
- Phenolic constituents in the fruits of Cinnamomum zeylanicum and their antioxidant activity: G.K. Jayaprakasha, et al.; J. Agric. Food Chem. 54, 1672 (2006)
- Influence of cranberry phenolics on glucan synthesis by glucosyltransferases and Streptococcus mutans acidogenicity: S. Gregoire, et al.; J. Appl. Microbiol. 103, 1960 (2007)
- Influence of cranberry proanthocyanidins on formation of biofilms by Streptococcus mutans on saliva-coated apatitic surface and on dental caries development in vivo: H. Koo, et al.; Caries Res. 44, 116 (2010)
- Doubly linked, A-type proanthocyanidin trimer and other constituents of Ixora coccinea leaves and their antioxidant and antibacterial properties: T.O. Idowu, et al.; Phytochemistry 71, 2092 (2010)
- Inhibition of viral RNA synthesis in canine distemper virus infection by proanthocyanidin A2: L. Gallina, et al.; Antiviral Res. 92, 447 (2011)
- Flavonoids from Lindera glauca blume as low-density lipoprotein oxidation inhibitors: G.W. Huh, et al.; Nat. Prod. Res. 28, 831 (2014)
- Procyanidins from the stem wood of Machilus japonica and their inhibitory effect on LDL oxidation: H.J. Park, et al.; Arch. Pharm. Res. 37, 1403 (2014)
- Chemical composition, antioxidant and antinociceptive properties of Litchi chinensis leaves: R.C. Castellain, et al.; J. Pharm. Pharmacol. 66, 1796 (2014)
- Preventive effects of procyanidin A2 on glucose homeostasis, pancreatic and duodenal homebox 1, and glucose transporter 2 gene expression disturbance induced by bisphenol A in male mice: A. Ahangarpour, et al.; J. Physiol. Pharmacol. 67, 243 (2016)






