Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
AdipoGen Life Sciences
SB202190
As low as
35
CHF
CHF 35.00
In stock
Only %1 left
AG-CR1-0028-M0011 mgCHF 35.00
AG-CR1-0028-M0055 mgCHF 55.00
AG-CR1-0028-M02525 mgCHF 95.00
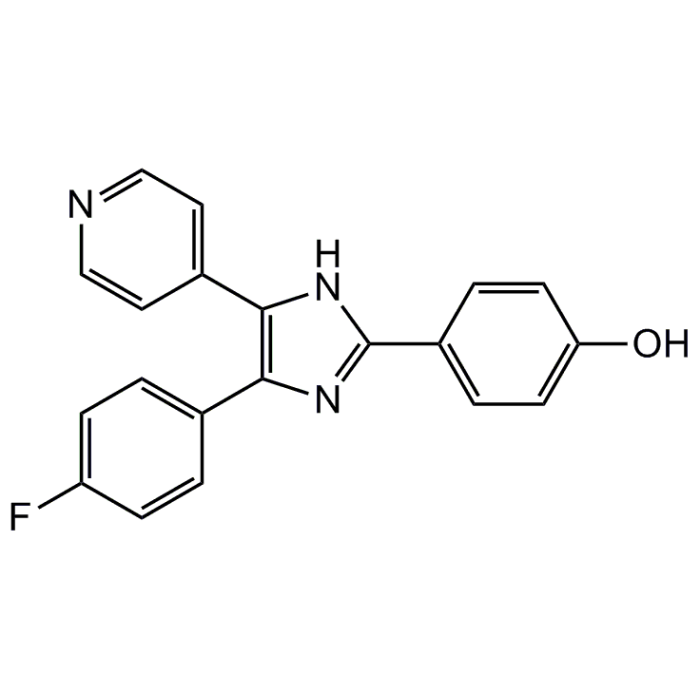
| Product Details | |
|---|---|
| Synonyms | FHPI; 4-(4-Fluorophenyl)-2-(4-hydroxyphenyl)-5-(4-pyridyl)-1H-imidazole |
| Product Type | Chemical |
| Properties | |
| Formula |
C20H14FN3O |
| MW | 331.3 |
| CAS | 152121-30-7 |
| Purity Chemicals | ≥97% (HPLC) |
| Appearance | Off-white solid. |
| Solubility | Soluble in methanol, DMSO or acetone; slightly soluble in ethyl acetate. |
| Identity | Determined by 1H-NMR. |
| InChi Key | QHKYPYXTTXKZST-UHFFFAOYSA-N |
| Smiles | OC1=CC=C(C=C1)C1=NC(=C(N1)C1=CC=NC=C1)C1=CC=C(F)C=C1 |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice | Protect from light. |
| Use/Stability | Stable for at least 2 years after receipt when stored at -20°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
- Potent and cell permeable p38 MAP kinase inhibitor [1, 2].
- Apoptosis inducer [3].
- Inhibits p38α and β, but not γ and δ isoforms [4, 5].
- Does not inhibit ERK2 or other members of the MAP kinase family or their upstream activators [6].
- JNK activator [7].
- Autophagic vacuole inducer [8]
Product References
- A protein kinase involved in the regulation of inflammatory cytokine biosynthesis: J.C. Lee, et al.; Nature 372, 739 (1994)
- SB202190, a selective inhibitor of p38 mitogen-activated protein kinase, is a powerful regulator of LPS-induced mRNAs in monocytes: C.L Manthey, et al.; J. Leukoc. Biol. 64, 409 (1998)
- Induction of apoptosis by SB202190 through inhibition of p38beta mitogen-activated protein kinase: S. Nemoto, et al.; J. Biol. Chem. 273, 16415 (1998)
- Characterization of the structure and function of a new mitogen-activated protein kinase (p38beta): Y. Jiang, et al.; J. Biol. Chem. 271, 17920 (1996)
- The structural basis for the specificity of pyridinylimidazole inhibitors of p38 MAP kinase: K.P. Wilson, et al.; Chem. Biol. 4, 423 (1997)
- A single amino acid substitution makes ERK2 susceptible to pyridinyl imidazole inhibitors of p38 MAP kinase: T. Fox, et al.; Protein Sci. 7, 2249 (1998)
- Activation of c-Jun N-terminal kinase (JNK) by widely used specific p38 MAPK inhibitors SB202190 and SB203580: a MLK-3-MKK7-dependent mechanism: H. Muniyappa & K.C. Das; Cell. Signal. 20, 675 (2008)
- SB202190-induced cell type-specific vacuole formation and defective autophagy do not depend on p38 MAP kinase inhibition: MB. Menon, et al.; PLos One 6, e23054 (2011)
- A preliminary study on the proinflammatory mechanisms of Treponema pallidum outer membrane protein Tp92 in human macrophages and HMEC-1 cells: X. Luo, et al.; Microb. Pathog. 110, 176 (2017)
- Candida albicans infection disturbs redox homeostasis system and induces reactive oxygen species accumulation for epithelial cell death: T. Ren, et al.; FEMS Yeast Res. ahead of print (2019)
- Signaling pathways involved in anti-inflammatory effects of Pulsed Electromagnetic Field in microglial cells: S. Merighi, et al.; Cytokine 125, 154777 (2020)





