Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
BioViotica
Chetomin
As low as
205
CHF
CHF 205.00
In stock
Only %1 left
BVT-0161-M0011 mgCHF 205.00
BVT-0161-M0055 mgCHF 710.00
| Product Details | |
|---|---|
| Synonyms | CTM; Chaetomin; NSC289491; BRN0077366 |
| Product Type | Chemical |
| Properties | |
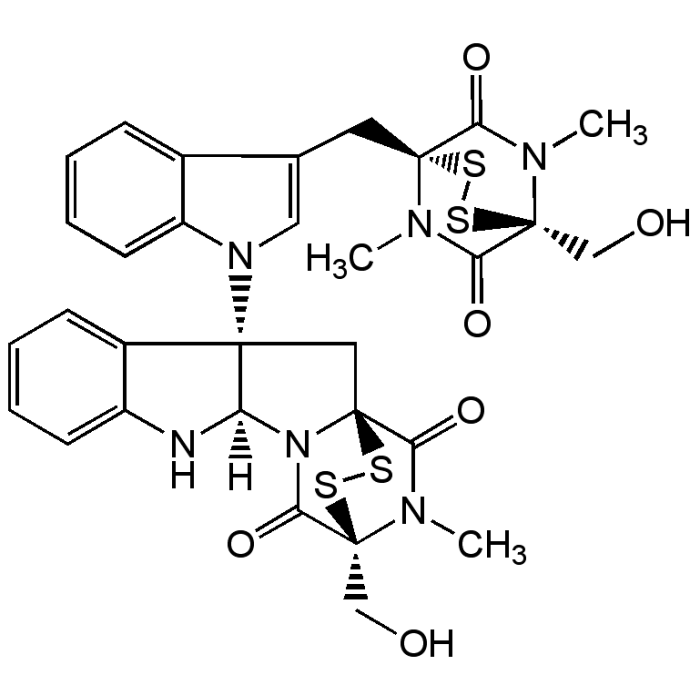
| Formula |
C31H30N6O6S4 |
| MW | 710.9 |
| CAS | 1403-36-7 |
| Source/Host Chemicals | Isolated from Chaetomium sp. |
| Purity Chemicals | ≥90% (HPLC) |
| Appearance | Off-white to fawn solid. |
| Solubility | Soluble in DMSO, ethyl acetate or pyridine; fairly soluble in methanol or 100% ethanol; insoluble in water. |
| Identity | Determined by 1H-NMR. |
| Declaration | Manufactured by BioViotica. |
| InChi Key | ZRZWBWPDBOVIGQ-OKMJTBRXSA-N |
| Smiles | [H][C@]12NC3=CC=CC=C3[C@]1(C[C@]13SS[C@@](CO)(N(C)C1=O)C(=O)N23)N1C=C(C[C@]23SS[C@@](CO)(N(C)C2=O)C(=O)N3C)C2=CC=CC=C12 |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice |
Protect from moisture. Protect from light when in solution. |
| Use/Stability |
Stable for at least 1 year after receipt when stored at -20°C. After reconstitution protect from light at -20°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
- Anticancer antibiotic.
- Inhibitor of HIF-1 formation by disrupting the binding of p300 to both HIF-1α and HIF-2α.
- Histone H3K9 methyltransferases G9a and Suv39h inhibitor.
- Tumor growth inhibitor.
- Angiogenesis inhibitor.
- Potent immunosuppressor.
- Antibacterial and antifungal.
- Strong inducer of retinal pigment epithelium (RPE).
Product References
- The structure of chetomin: A.G. McInnes, et al.; JACS 98, 6741 (1976)
- Effects of chetomin on growth and acidic fermentation products of rumen bacteria: W.C. Jen and G.A. Jones; Can. J. Microbiol. 29, 1399 (1983)
- Immunomodulatory constituents from an Ascomycete, Chaetomiumseminudum: H. Fujimoto, et al.; J. Nat. Prod. 67, 98 (2004)
- Small molecule blockade of transcriptional coactivation of the hypoxia-inducible factor pathway: A.L. Kung, et al.; Cancer Cell 6, 33 (2004)
- Effects of HIF-1 inhibition by chetomin on hypoxia-related transcription and radiosensitivity in HT 1080 human fibrosarcoma cells: A. Staab, et al.; BMC Cancer 7, 213 (2007)
- Epidithiodiketopiperazines block the interaction between hypoxia-inducible factor-1α (HIF-1α) and p300 by a zinc ejection mechanism: K.M. Cook, et al.; J. Biol. Chem. 284, 26831 (2009)
- Direct inhibition of hypoxia-inducible transcription factor complex with designed dimeric epidithiodiketopiperazine: K. M. Wang, et al.; JACS 131, 18078 (2009)
- Up-regulation of pro-inflammatory genes as adaptation to hypoxia in MCF-7 cells and in human mammary invasive carcinoma microenvironment: M. Tafani, et al.; Cancer Sci. 101, 1014 (2010)
- Inhibition of histone H3K9 methyltransferases by gliotoxin and related epipolythiodioxopiperazines: M. Takahashi, et al., J. Antibiot. 65, 263 (2012)
- Epidithiodiketopiperazines (ETPs) exhibit in vitro antiangiogenic and in vivo antitumor activity by disrupting the HIF-1α/p300 complex in a preclinical model of prostate cancer: K.M. Reece, et al.; Mol. Cancer 13, 91 (2014)
- Small-molecule–directed, efficient generation of retinal pigment epithelium from human pluripotent stem cells: J. Maruotti, et al.; PNAS 112, 10950 (2015)
- Chetomin, targeting HIF-1a/p300 complex, exhibits antitumour activity in multiple myeloma: E. Viziteu, et al.; Br. J. Canc. 114, 519 (2016)
- Chetomin induces apoptosis in human triple-negative breast cancer cells by promoting calcium overload and mitochondrial dysfunction: J. Dewangan, et al.; BBRC 495, 1915 (2018)
- Screening of a growth inhibitor library of sarcoma cell lines to identify potent anti-cancer drugs: Z. Qiao & T. Kondo; Electrophoresis 63, 1 (2019)
- Chetomin, a Hsp90/HIF1a pathway inhibitor, effectively targets lung cancer stem cells and non-stem cells: S. Min, et al.; Cancer Biol. Ther. 21, 698 (2020)