Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
Chemodex
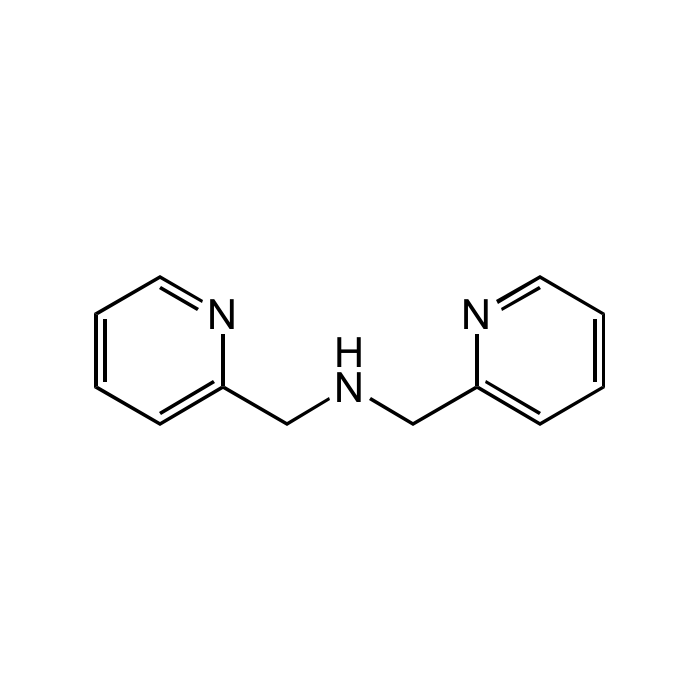
Di-(2-picolyl)amine
| Product Details | |
|---|---|
| Synonyms | Dipicolylamine; DPA; Bis(2-pyridylmethyl)amine; 2,2'-Bis(pyridylmethyl)amine; NSC 176070 |
| Product Type | Chemical |
| Properties | |
| Formula |
C12H13N3 |
| MW | 199.25 |
| CAS | 1539-42-0 |
| Source/Host Chemicals | Synthetic |
| Purity Chemicals | ≥95% (GC) |
| Appearance | Yellow to orange liquid. |
| Solubility | Sligthly soluble in water. |
| Identity | Determined by NMR. |
| Declaration | Manufactured by Chemodex. |
| Other Product Data |
Click here for Original Manufacturer Product Datasheet |
| InChi Key | KXZQYLBVMZGIKC-UHFFFAOYSA-N |
| Smiles | C(NCC1=CC=CC=N1)C1=CC=CC=N1 |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice |
Keep cool and dry. Protect from light and moisture. |
| Use/Stability | Stable for at least 2 years after receipt when stored at -20°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Building block for synthesis. DPA is a secondary amine with two picolyl substituents. The compound is a tridentate ligand in coordination chemistry and commonly used to produce Zn-based chemosensors/probes, such as Zinpry. As a tridentate ligand this compound provides three nitrogen donors that affords good selectivity for Zn2+ over biologically relevant metals such as Na+, K+, Mg2+ and Ca2+, and leaves coordination sites free for anion binding. The amino nitrogen of the DPA group is a good candidate as an electron donor in either photoinduced electron transfer or photoinduced charge transfer (PET or PCT) sensors. Zn(II)–DPA complexes are widely used in anion recognition and sensing.
(1) E.M. Nolan & S.J. Lippard; Inorg. Chem. 43, 8310 (2004) | (2) N.C. Lim & C. Bruckner; Chem. Commun. 2004, 1094 (2004) | (3) H.-W. Lee, et al.; Bull. Korean Chem. Soc. 28, 855 (2007) | (4) L. Xue, et al.; Inorg. Chem. 47, 4310 (2008) | (5) T. Sakamoto, et al.; Chem. Commun. 2009, 141 (2009) | (6) L. You, et al.; Nat. Chem. 3, 943 (2011) | (7) H.T. Ngo, et al.; Chem. Soc. Rev. 41, 4928 (2012)