Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
Chemodex
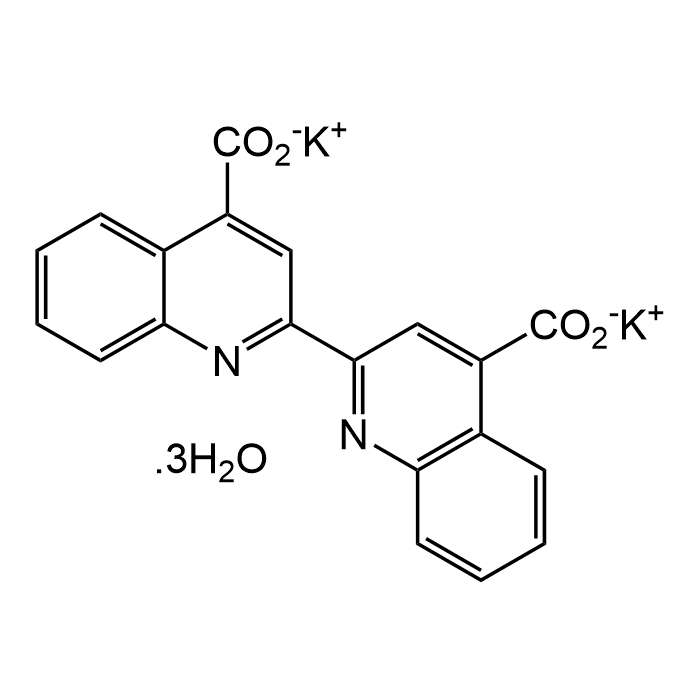
2,2'-Bicinchoninic acid dipotassium salt trihydrate
| Product Details | |
|---|---|
| Synonyms | BQC; Potassium [2,2'-biquinoline]-4,4'-dicarboxylate |
| Product Type | Chemical |
| Properties | |
| Formula | C20H10K2N2O4 . 3H2O |
| MW | 474.55 |
| CAS | 207124-63-8 |
| Source/Host Chemicals | Synthetic |
| Purity Chemicals | ≥98% |
| Appearance | Off white to light red solid. |
| Solubility | Soluble in water. |
| Identity | Determined by 1H-NMR. |
| Declaration | Manufactured by Chemodex. |
| Other Product Data |
Click here for Original Manufacturer Product Datasheet |
| InChi Key | MUCQANVZZZELIW-UHFFFAOYSA-L |
| Smiles | [K+].[K+].[H]O[H].[H]O[H].[H]O[H].[O-]C(=O)c1cc(nc2ccccc12)-c3cc(C([O-])=O)c4ccccc4n3 |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +20°C |
| Long Term Storage | +20°C |
| Handling Advice | Protect from light and moisture. |
| Use/Stability | Stable for at least 2 years after receipt when stored at RT. |
| Documents | |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
2,2´-Biquinoline-4,4´-dicarboxylic acid dipotassium salt (BQC) is a water soluble biquinoline-based ligand. The oxidation of alcohols to the corresponding aldehydes and ketones, which can be performed by a variety of methods, remains one of the most important reactions in organic synthesis. BOC is used in the aerobic oxidation of alcohols (primary, secondary and benzylic) in the presence of palladium catalysts. The catalytic system composed of CuCl2 and BQC was found to be highly efficient for the selective oxidation of secondary benzylic, allylic and propargylic alcohols to the corresponding ketones, with aqueous t-butyl hydroperoxide under phase-transfer catalysis conditions. The catalytic system is stable and can be recycled and reused several times without loss of activity. Since the reaction solvent is only water and the oxidant is air, it may be used as a solvnt-free green alternative to the traditional methods of oxidation. BOC has also been used as an aqueous-phase catalyst for the hydrogenation of nitriles. As a complexing agent, BOC assists in the identification, extraction, and quantification of metal residues. BOC has been used for the determination of copper in wine. It binds to Cu(I) and forms a water-soluble complex that absorbs strongly at 562 nm.
(1) G. Ferguson & A.N. Ajjou; Tetrahed. Lett. 44, 9139 (2003) | (2) A.N. Ajjou & J.-L. Pinet; J. Mol. Catal. A: Chem. 214, 203 (2004) | (3) B.P. Buffin, et al.; J. Mol. Catal. A: Chem. 225, 111 (2005) | (4) J. Boudreaz, et al.; Tetrahed. Lett. 47, 1695 (2006) | (5) A.N. Ajjou & G. Ferguson; Tetrahed. Lett. 47, 3719 (2006) | (6) D.M. Chisholm & J.S. McIndoe; Dalton Trans. 30, 3933 (2008) | (7) A. Rahman, et al.; Chin. Chem. Lett. 22, 691 (2011) | (8) A.N. Ajjou & A. Robichaud; Appl. Organometall. Chem. 32, e4547 (2018) | (9) K. Krommyda, et al.; Catal. Lett. 149, 1250 (2019) | (10) N. Kontoudakis, et al.; Austr. J. Grape Wine Res. 26, 121 (2020)





