Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
Chemodex
Celecoxib
As low as
39
CHF
CHF 39.00
In stock
Only %1 left
CDX-C0439-M01010 mgCHF 39.00
CDX-C0439-M05050 mgCHF 90.00
CDX-C0439-G0011 gCHF 155.00
| Product Details | |
|---|---|
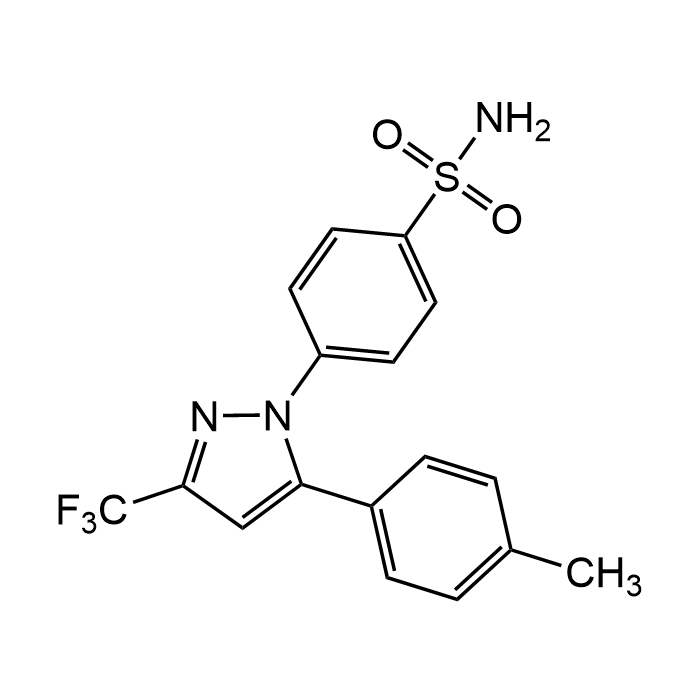
| Synonyms | 4-[5-(4-Methylphenyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl]benzenesulfonamide; SC 58635; YM 177 |
| Product Type | Chemical |
| Properties | |
| Formula | C17H14F3N3O2S |
| MW | 381.37 |
| CAS | 169590-42-5 |
| RTECS | DB2944937 |
| Source/Host Chemicals | Synthetic |
| Purity Chemicals | ≥98% (HPLC) |
| Appearance | White to beige powder. |
| Solubility | Soluble in DMSO (15mg/ml), DMF (25mg/ml) or ethanol (25mg/ml). Insoluble in water. |
| Identity | Determined by 1H-NMR. |
| Declaration | Manufactured by Chemodex. |
| Other Product Data |
Click here for Original Manufacturer Product Datasheet |
| InChi Key | RZEKVGVHFLEQIL-UHFFFAOYSA-N |
| Smiles | O=S(=O)(N)C1=CC=C(C=C1)N2N=C(C=C2C=3C=CC(=CC3)C)C(F)(F)F |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +20°C |
| Long Term Storage | +4°C |
| Handling Advice | Protect from light and moisture. |
| Use/Stability | Stable for at least 2 years after receipt when stored at +4°C. |
| Documents | |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
Celecoxib is a nonsteroidal anti-inflammatory drug (NSAID) and a highly selective cyclooxygenase-2 (COX-2) inhibitor. By preferentially inhibiting COX-2 over COX-1, celecoxib suppresses the formation of pro-inflammatory prostaglandins while minimizing effects on gastric COX-1 activity. It is widely used as a pharmacological tool in inflammation, pain, cancer, and cardiovascular research. In addition to its well-established anti-inflammatory mechanism, celecoxib has been shown to exert COX-2-independent biological effects, including induction of apoptosis, modulation of Bcl-2 family proteins, and regulation of ion channel activity. These properties make celecoxib a widely used research tool in inflammation, oncology, and cell-death signaling studies.
Product References
[1] M.M. Goldenberg; Clin. Ther. 21, 1497 (1999) (Review) | [2] N.M. Davies, et al.; Clin. Pharmacokinet. 38, 225 (2000) (Review) | [3] D. Clemett & K.L. Goa; Drugs 59, 957 (2000) (Review) | [4] I.A. Mardini & G.A. Fitzgerald; Mol. Interv. 1, 30 (2001) | [5] A.T. Koki & J.L. Masferrer; Cancer Control 9, 28 (2002) (Review) | [6] V. Jendrossek, et al.; FASEB J. 17, 1547 (2003) | [7] K. Kismet, et al.; Cancer Detect. Prev. 28, 127 (2004) (Review) | [8] D. Jiang, et al.;Int. J. Mol. Sci. 11, 4106 (2010) | [9] K. Antoniou, et al.; Expert Opin. Pharmacother. 8, 1719 (2007) (Review) | [10] L.L. Winfield & F. Payton-Stewart; Future Med. Chem. 4, 361 (2012) (Review) | [11] V. Jendrossek; Cancer Lett. 332, 313 (2013) (Review) | [12] R.V. Frolov & S. Singh; Eur. J. Pharmacol. 730, 61 (2014) (Review) | [13] A.F. Khafaga, et al.; Nanomedicine 16, 1691 (2021) (Review)





