Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
Chemodex
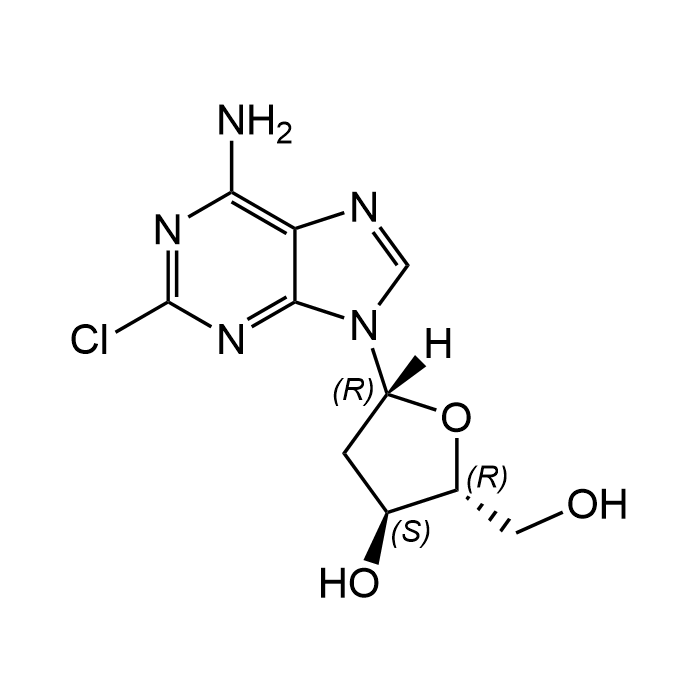
2-Chloro-2'-deoxyadenosine
As low as
32
CHF
CHF 32.00
In stock
Only %1 left
CDX-C0805-M01010 mgCHF 32.00
CDX-C0805-M05050 mgCHF 135.00
| Product Details | |
|---|---|
| Synonyms | CdA; Cladribine; 2-Chlorodeoxyadenosine; 2-Chloro-2′-deoxy-β-adenosine; NSC 105014-F; RWJ 26251 |
| Product Type | Chemical |
| Properties | |
| Formula | C10H12ClN5O3 |
| MW | 285.69 |
| CAS | 4291-63-8 |
| RTECS | AU7357560 |
| Source/Host Chemicals | Synthetic |
| Purity Chemicals | ≥98% (HPLC) |
| Appearance | White to off-white powder. |
| Solubility | Soluble in DMSO (10mg/ml) or DMF (10mg/ml). |
| Identity | Determined by 1H-NMR. |
| Declaration | Manufactured by Chemodex. |
| Other Product Data |
Click here for Original Manufacturer Product Datasheet |
| InChi Key | PTOAARAWEBMLNO-KVQBGUIXSA-N |
| Smiles | NC1=C2C(N(C=N2)[C@@H]3O[C@H](CO)[C@@H](O)C3)=NC(Cl)=N1 |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +20°C |
| Long Term Storage | +4°C |
| Handling Advice | Protect from light and moisture. |
| Use/Stability | Stable for at least 2 years after receipt when stored at +4°C. |
| Documents | |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
2-Chloro-2′-deoxyadenosine is a synthetic anticancer purine nucleoside analog commonly used in biochemical and cellular research to study nucleoside metabolism, DNA synthesis, and apoptosis pathways. Due to its resistance to adenosine deaminase–mediated degradation, this compound exhibits enhanced stability in biological systems, making it a valuable tool for mechanistic studies involving DNA replication and repair. Cladribine is cytotoxic to resting or proliferating lymphocytes. It inhibits RNA synthesis and induces DNA strand breaks in resting human peripheral blood lymphocytes. Cladribine induces cell cycle arrest at the G1 phase and apoptosis in U266, RPMI-8226, and MM.1S multiple myeloma cells in a concentration-dependent manner. It reduces tumor growth in HT-29 colon cancer, RL lymphoma, and RPMI-8226 multiple myeloma mouse xenograft models.
Product References
[1] D.A. Carson, et al.; Blood 62, 737 (1983) | [2] S. Seto, et al.; J. Clin. Invest. 75, 377 (1985) | [3] E. Beutler; Lancet 340, 952 (1992) (Review) | [4] A. Gorski, et al.; Immunopharmacol. 26, 197 (1993) | [5] S. Spurgeon, et al.; Expert Opin. Investig. Drugs 18, 1169 (2009) (Review) | [6] J. Ma, et al.; BMC Cancer 11, 255 (2011) | [7] T.P. Leist & R. Weissert; Clin. Neuropharmacol. 34, 28 (2011) (Review) | [8] R. Hermann, et al.; Clin. Pharmacokinet. 60, 1509 (2021) (Review) | [9] R. Hermann, et al.; Clin. Pharmacokinet. 61, 167 (2022) (Review)





