Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
Chemodex
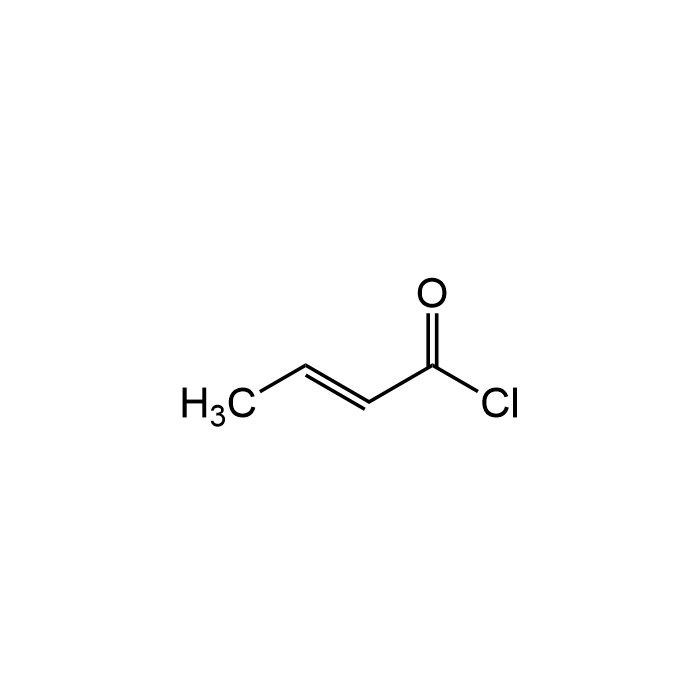
Crotonyl chloride
As low as
77
CHF
CHF 77.00
In stock
Only %1 left
CDX-C0882-G0055 gCHF 77.00
CDX-C0882-G02525 gCHF 122.00
CDX-C0882-40254 x 25 gCHF 367.00
| Product Details | |
|---|---|
| Synonyms | (E)-Crotonoyl chloride; trans-Crotonoyl chloride; (2E)-2-Butenoyl chloride; trans-2-Butenoyl chloride |
| Product Type | Chemical |
| Properties | |
| Formula | C4H5ClO |
| MW | 104.53 |
| CAS | 625-35-4 |
| Source/Host Chemicals | Synthetic |
| Purity Chemicals | ≥90% (GC) |
| Appearance | Colorless to light yellow liquid. |
| Solubility | Soluble in chloroform. Insoluble in water. |
| Identity | Determined by 1H-NMR. |
| Declaration | Manufactured by Chemodex. |
| Other Product Data |
Click here for Original Manufacturer Product Datasheet |
| InChi Key | RJUIDDKTATZJFE-NSCUHMNNSA-N |
| Smiles | C(\C(Cl)=O)=C/C |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | +4°C |
| Handling Advice | Protect from light and moisture. |
| Use/Stability | Stable for at least 2 years after receipt when stored at +4°C. |
| Documents | |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
Crotonyl chloride is a reactive acyl chloride commonly used as a building block or intermediate in organic synthesis and biochemical reagent preparation. It serves as an important intermediate for the introduction of crotonyl functional groups in small molecules, peptides, and proteins, and is frequently used in studies involving post-translational modifications such as lysine crotonylation. This reagent is well suited for chemical synthesis applications, including acylation reactions and the preparation of crotonylated substrates for epigenetics and chromatin biology research.
Product References
[1] D.K. Lum, et al.; J. Mol. Struct. 50, 1 (1978) | [2] R.K. Gupta, et al.; Spectr. Acta A Mol. Spectr. 45, 595 (1989) | [3] J.P. Murphy, et al.; Tetrahedr. 53, 11827 (1997) | [4] M. Zakeri, et al.; J. Chem. Sci. 125, 731 (2013) | [5] E. Catıker, et al.; Polym. Bull. 75, 47 (2018) | [6] M. El-Khateeb, et al.; J. Chem. Sci. 132, 22 (2020)





