Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
Chemodex
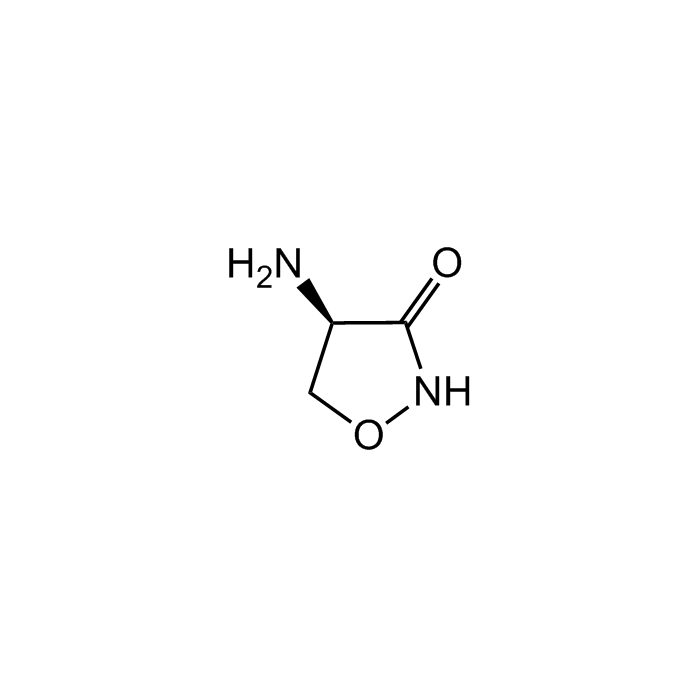
D-Cycloserine
| Product Details | |
|---|---|
| Synonyms | (R)-4-Amino-3-isoxazolidone; D-4-amino-3-isoxazolidone; D-Oxamycin; Seromycin; K300, NJ-21; (+)-Cycloserine; α-Cycloserine; (R)-Cycloserine; NSC 76029; NSC 154851 |
| Product Type | Chemical |
| Properties | |
| Formula |
C3H6N2O2 |
| MW | 102.09 |
| CAS | 68-41-7 |
| RTECS | NY2975000 |
| Purity Chemicals | ≥98% (TLC) |
| Appearance | White to off-white powder. |
| Solubility | Soluble in water. |
| Identity | Determined by 1H-NMR. |
| Declaration | Manufactured by Chemodex. |
| Other Product Data |
Click here for Original Manufacturer Product Datasheet |
| InChi Key | DYDCUQKUCUHJBH-UWTATZPHSA-N |
| Smiles | O=C1NOC[C@H]1N |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice | Protect from light and moisture. |
| Use/Stability | Stable for at least 2 years after receipt when stored at -20°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
D-cycloserine (DCS) is an antibiotic, inhibiting cell-wall biosynthesis in bacteria. It is a tuberculostatic agent, that inhibits L-alanine racemase and D-alanine:D-alanine ligase, enzymes essential to peptidoglycan synthesis and bacterial cell wall formation. Formulations containing DCS have been used as second-line agents to treat drug resistant tuberculosis. A reason for limited use of this drug is the neurological side effects it causes, since it is able to penetrate into the central nervous system (CNS). DCS acts as a glutamatergic partial N-methyl-D-aspartate (NMDA) agonist. It selectively binds at the glycine-binding site of the NMDA receptor and enable the opening of the NMDA channel. DCS has the ability to improve memory retention in senescence-accelerated mice which exhibit impaired learning and memory.
(1) W.F. Hood, et al.; Neurosci. Lett. 98, 91 (1989) | (2) G.B. Watson, et al.; Brain Res. 510, 158 (1990) | (3) J.F. Flood, et al.; Eur. J. Pharmacol. 221, 249 (1992) | (4) H. Baran, et al.; Brain Res. 652, 195 (1994) | (5) Z. Feng & R.G. Barletta; Antimicrob. Agents Chemother. 47, 283 (2003) | (6) J.A. Caminero, et al.; Lancet Infect. Dis. 10, 621 (2010) | (7) W. Hong, et al.; Expert Opin. Ther. Targets 18, 691 (2014) (Review) | (8) S. Schade & W. Paulus; Int. J. Neuropsychopharmacol. 19, pyv102 (2016) (Review)





