Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
Chemodex
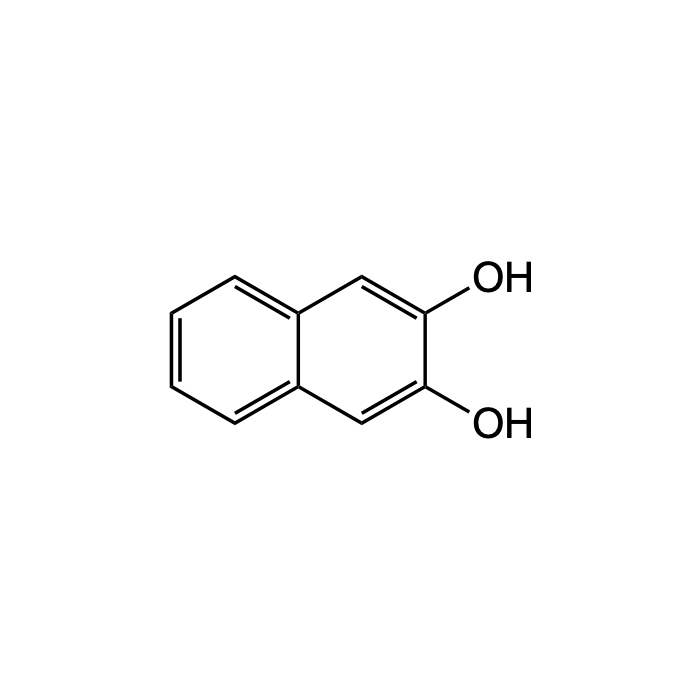
2,3-Dihydroxy-naphthalin
| Product Details | |
|---|---|
| Synonyms | 2,3-DHN; 2,3-Naphthalenediol; 2,3-Dihydroxy-naphthalin; 2,3-Naphthalindiol |
| Product Type | Chemical |
| Properties | |
| Formula | C10H8O2 |
| MW | 160.17 |
| CAS | 92-44-4 |
| RTECS | QJ4750000 |
| Purity Chemicals | ≥98% (HPLC) |
| Appearance | White to faint beige powder or crystals. |
| Solubility | Soluble in DMSO or methanol (1mg/ml). |
| Identity | Determined by 1H-NMR. |
| Declaration | Manufactured by Chemodex. |
| Other Product Data |
Click here for Original Manufacturer Product Datasheet |
| InChi Key | JRNGUTKWMSBIBF-UHFFFAOYSA-N |
| Smiles | Oc1cc2ccccc2cc1O |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +20°C |
| Long Term Storage | +20°C |
| Handling Advice |
Keep under inert gas. Very hygroscopic. |
| Use/Stability | Stable for at least 2 years after receipt when stored at RT. |
| Documents | |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
2,3-Dihydroxynaphthalene (2,3-DHN) is a naturally occurring catechol. 2,3-Dihydroxynaphthalene (2,3-DHN) can be used as a building block, starting material or reagent in organic synthesis to create more complex chemical compounds. The hydroxyl groups in 2,3-DHN can undergo various chemical reactions, such as esterification, oxidation, or substitution reactions, to produce a wide range of derivatives. 2,3-Dihydroxynaphthalene displays the ability to function as either an electron donor or acceptor in redox reactions. It also serves as an electron transfer agent in enzymatic reactions. 2,3-DHN and its derivatives can be used in the synthesis of dyes and pigments. 2,3-DHN and its derivatives have been shown to inhibit certain enzymes and have potential anti-tumor properties. 2,3-DHN is a metal chelators.
(1) J.C. DiNardo, et al.; Toxicol. Appl. Pharmacol. 78, 163 (1985) | (2) P.K. Tarafder, et al.; Talanta 41, 1345 (1994) | (3) R. Shiman, et al.; J. Cell Biol. 236, 24637 (1994) | (4) S. Bhownik & U. Maitra; Chem. Commun. 48, 4624 (2012) | (5) J. Feng, et al.; Dalton Trans. 41, 8697 (2012) | (6) L.M. Laglera, et al.; Anal. Chem. 85, 2486 (2013) | (7) M. Burton, et al.; Bioorg. Med. Chem. 26, 4841 (2018)





