Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
Chemodex
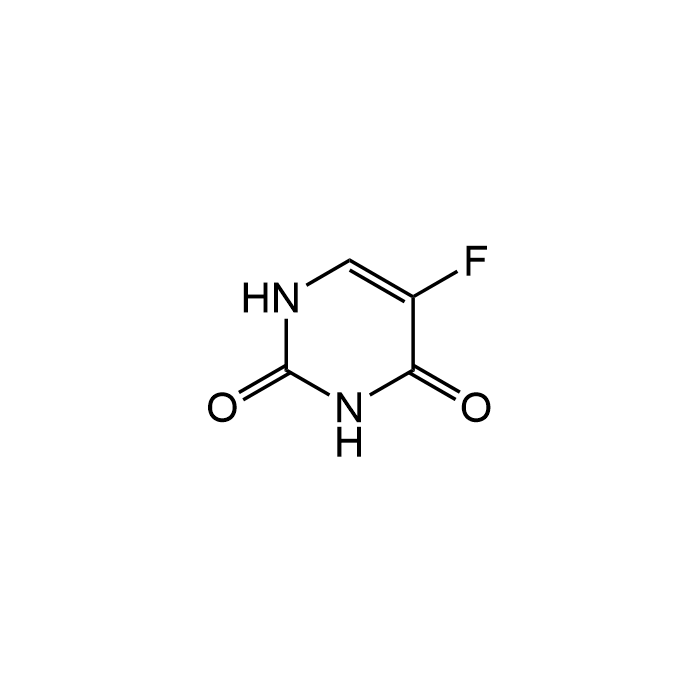
Fluorouracil
As low as
116
CHF
CHF 116.00
In stock
Only %1 left
CDX-F0140-G02525 gCHF 116.00
CDX-F0140-40254 x 25 gCHF 348.00
| Product Details | |
|---|---|
| Synonyms | 5-Fluorouracil; 2,4-Dihydroxy-5-fluoropyrimidine; NSC 19893; Ro 2-9757; 5-FU; 5-Fluoro-2,4-pyrimidinedione; |
| Product Type | Chemical |
| Properties | |
| Formula | C4H3FN2O2 |
| MW | 130.08 |
| CAS | 51-21-8 |
| RTECS | YR0350000 |
| Source/Host Chemicals | Synthetic |
| Purity Chemicals | ≥97% (UV) |
| Appearance | White to off-white powder. |
| Solubility | Soluble in DMSO (20mg/ml) or DMF (20mg/ml). |
| Identity | Determined by 1H-NMR. |
| Declaration | Manufactured by Chemodex. |
| Other Product Data |
Click here for Original Manufacturer Product Datasheet |
| InChi Key | GHASVSINZRGABV-UHFFFAOYSA-N |
| Smiles | O=C1NC=C(F)C(=O)N1 |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +20°C |
| Long Term Storage | +4°C |
| Handling Advice | Protect from light and moisture. |
| Use/Stability | Stable for at least 2 years after receipt when stored at +4°C. |
| Documents | |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
Fluorouracil (5-FU) is a fluorinated pyrimidine analog that disrupts nucleic acid synthesis through multiple intracellular mechanisms. Following cellular uptake, 5-FU is metabolically activated to several cytotoxic nucleotide derivatives that interfere with both DNA and RNA metabolism. One major active metabolite, 5-fluoro-2′-deoxyuridine-5′-monophosphate (FdUMP), forms a stable ternary complex with thymidylate synthase (TS) and the folate cofactor 5,10-methylenetetrahydrofolate. This complex inhibits TS activity, leading to depletion of deoxythymidine monophosphate (dTMP) and subsequently reduced dTTP pools. The resulting imbalance in deoxynucleotide levels causes DNA replication stress, stalled replication forks, and DNA damage. In parallel, 5-FU is converted to 5-fluorouridine triphosphate (FUTP), which can be incorporated into RNA in place of uridine. This incorporation disrupts RNA processing, ribosomal assembly, and translation, impairing protein synthesis. Additionally, formation of 5-fluoro-2′-deoxyuridine triphosphate (FdUTP) allows misincorporation into DNA, triggering base excision repair pathways and further genomic instability. Collectively, these effects lead to cell cycle arrest, activation of DNA damage response pathways, and induction of apoptosis, particularly in rapidly proliferating cells.
Product References
[1] M.L. Eidinoff, et al.; Arch. Biochem. Biophys. 71, 274 (1957) | [2] B. Ardalan & R. Glazer; Cancer Treat. Rev. 8, 157 (1981) (Review) | [3] H.M. Pinedo & G.F. Peters; J. Clin. Oncol. 6, 1653 (1988) (Review) | [4] K. Ghoshal & S.T. Jacob; Biochem. Pharmacol. 53, 1569 (1997) (Review) | [5] P.M. McSheehy, et al.; Adv. Enzyme Regul. 40, 63 (2000) (Review) | [6] J.L. Arias; Molecules 13, 2340 (2008) (Review) | [7] F. Ghiringhelli & L. Apetoh; Biomed. J. 38, 111 (2015) (Review)





