Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
Chemodex
D-Luciferin 6'-methyl ether
As low as
187
CHF
CHF 187.00
In stock
Only %1 left
CDX-L0315-M0055 mgCHF 187.00
CDX-L0315-M01010 mgCHF 303.00
CDX-L0315-M02525 mgCHF 625.00
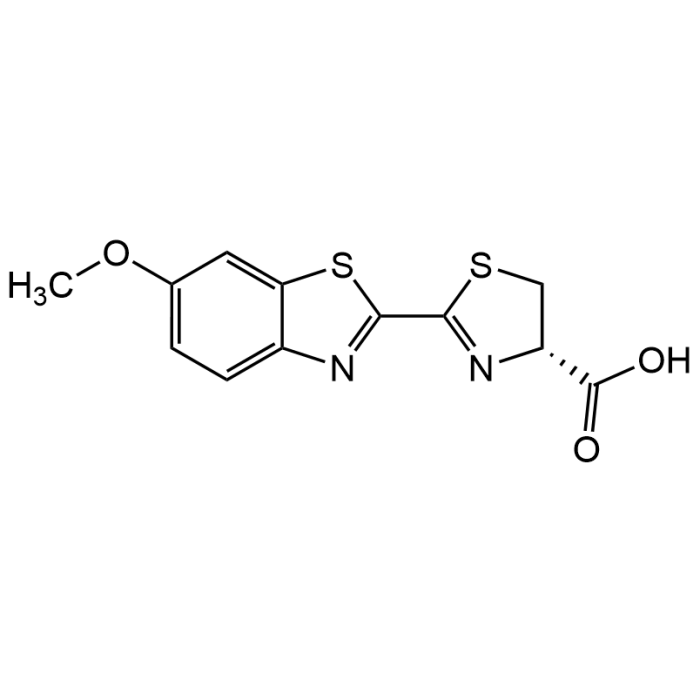
| Product Details | |
|---|---|
| Synonyms | 6′-Methoxyluciferin |
| Product Type | Chemical |
| Properties | |
| Formula | C12H10N2O3S2 |
| MW | 294.35 |
| CAS | 24404-90-8 |
| Source/Host Chemicals | Synthetic |
| Purity Chemicals | ≥98% (HPLC) |
| Appearance | White to faint yellow powder. |
| Solubility | Soluble in DMSO or methanol. |
| Identity | Determined by 1H-NMR. |
| Declaration | Manufactured by Chemodex. |
| Other Product Data |
Click here for Original Manufacturer Product Datasheet |
| InChi Key | ZTQKCGHSTKIWFW-MRVPVSSYSA-N |
| Smiles | OC([C@@H]1N=C(SC1)C(SC2=C3)=NC2=CC=C3OC)=O |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice | Protect from light and moisture. |
| Use/Stability | Stable for at least 2 years after receipt when stored at -20°C. |
| Documents | |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
D-Luciferin 6′-methyl ether is a chemically modified analog of D-luciferin in which the 6′-hydroxyl group is methylated. This structural modification significantly reduces productive oxidation by firefly luciferase while retaining enzyme binding affinity. As a result, the compound functions as a non-productive substrate analog that can competitively interfere with D-luciferin-dependent bioluminescence. D-Luciferin 6′-methyl ether is used in mechanistic enzymology and chemical biology to investigate luciferase substrate recognition, active-site interactions, and structure-activity relationships. It is also valuable for the evaluation of engineered or mutant luciferases with altered substrate specificity. Spectral Data: Excitation max: ~ 326nm, Emission max: ~430nm (accord. Lit).
Product References
[1] E.H. White, et al.; J. Org. Chem. 30, 2344 (1965) | [2] E.H. White, et al.; Bioorg. Cherm. 1, 92 (1971) | [3] O.A. Gandelman, et al.; J. Photochem. Photobiol. B. Biol. 19, 187 (1993) | [4] G. Meroni, et al.; Arkivoc 265-288 (2009) (Review)





