Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
Chemodex
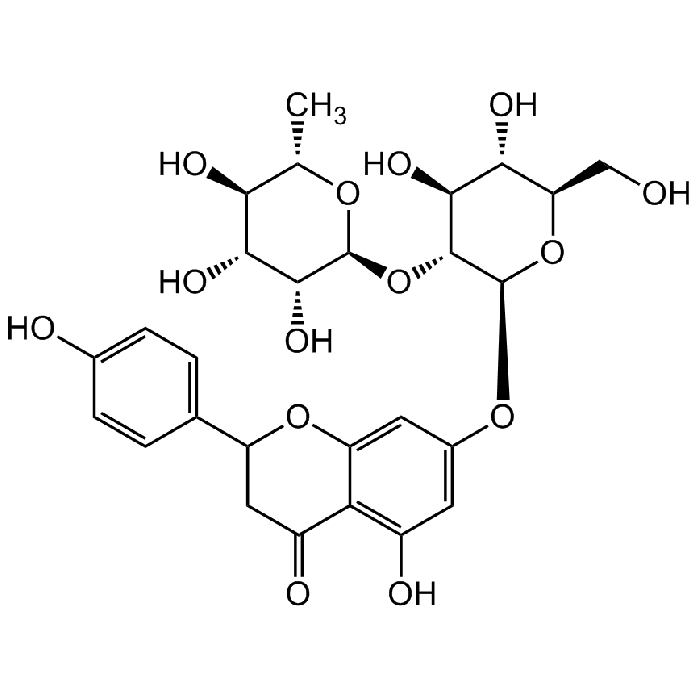
Naringin
| Product Details | |
|---|---|
| Synonyms | 4',5,7-Trihydroxyflavanone 7-rhamnoglucoside; Naringenin 7-neohesperidoside; Naringenine-7-rhamnosidoglucoside; Naringoside; NSC 5548 |
| Product Type | Chemical |
| Properties | |
| Formula |
C27H32O14 |
| MW | 580.54 |
| CAS | 10236-47-2 |
| RTECS | QN6340000 |
| Source/Host Chemicals | Plant |
| Purity Chemicals | ≥98% (HPLC) |
| Appearance | Light yellow to beige powder. |
| Solubility | Soluble in DMSO (10mg/ml), DMF (20mg/ml) or ethanol (1mg/ml). |
| Identity | Determined by 1H-NMR. |
| Declaration | Manufactured by Chemodex. |
| Other Product Data |
Click here for Original Manufacturer Product Datasheet |
| InChi Key | DFPMSGMNTNDNHN-JJLSSNRUSA-N |
| Smiles | OC1=C2C(OC(C3=CC=C(O)C=C3)CC2=O)=CC(O[C@@H]4O[C@H](CO)[C@@H](O)[C@H](O)[C@H]4O[C@@H]5O[C@@H](C)[C@H](O)[C@@H](O)[C@H]5O)=C1 |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +20°C |
| Long Term Storage | +4°C |
| Handling Advice | Protect from light and moisture. |
| Use/Stability | Stable for at least 2 years after receipt when stored at +4°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Naringin is a citrus-derived flavonoid (bitter principal). It undergoes extensive metabolism in the liver. It has a broad panel of properties, including anti-inflammatory, antioxidant, anticancer, antidiabetic, cardioprotective, bone regenerative and neuroprotective activities. Naringin acts by modulating several signaling pathway targets, activity is primarily attributed to its anti-inflammatory (via inhibiting recruitment of cytokines and inflammatory transcription factors) and antioxidant (via scavenging of free radicals, bolstering of endogenous antioxidant defense system and metal ion chelation) effects. It increases the expression of neurotrophic factor in dopaminergic neurons, providing neuroprotection.
(1) U. Fuhr, et al.; Br. J. Clin. Pharmacol. 35, 431 (1993) | (2) P.C. Ho & D.J. Saville; J. Pharm. Pharmaceut. Sci. 4, 217 (2001) | (3) S. Bharti, et al.; Planta Med. 80, 437 (2014) (Review) | (4) U.J. Jung, et al.; Exp. Neurobiol. 23, 124 (2014) (Review) | (5) M.A. Alam, et al.; Adv. Nutr. 5, 404 (2014) (Review) | (6) R. Chen, et al.; Pharm. Biol. 54, 3203 (2016) (Review)
- Anti-estrogenic and anti-aromatase activities of citrus peels major compounds in breast cancer: D.M. El-Kersh, et al.; Nature Sci. Rep. 11, 7121 (2021)





