Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
Chemodex
Naproxen Solution
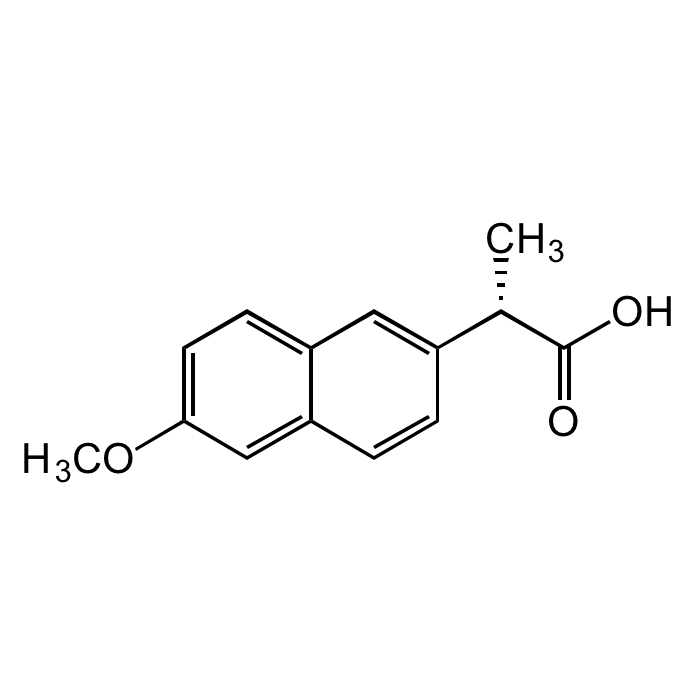
| Product Details | |
|---|---|
| Synonyms | (S)-Naproxen; (+)-Naproxen; (S)-(+)-6-Methoxy-α-methyl-2-naphthalene acetic acid; Equiproxen; Laraflex |
| Product Type | Chemical |
| Properties | |
| Formula |
C14H14O3 |
| MW | 230.26 |
| CAS | 22204-53-1 |
| RTECS | UF5275000 |
| Source/Host Chemicals | Synthetic. |
| Purity Chemicals | ≥98% (HPLC) |
| Appearance | Liquid. 1mg/ml in methanol. |
| Solubility | Soluble in aqueous buffers. |
| Identity | Determined by 1H-NMR. |
| Declaration | Manufactured by Chemodex. |
| Other Product Data |
Click here for Original Manufacturer Product Datasheet |
| InChi Key | CMWTZPSULFXXJA-VIFPVBQESA-N |
| Smiles | C[C@H](C(O)=O)C1=CC2=CC=C(OC)C=C2C=C1 |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | +4°C |
| Handling Advice | Protect from light and moisture. |
| Use/Stability | Stable for at least 2 years after receipt when stored at +4°C. |
| Documents | |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Naproxen is a potent, non-steroidal, non-selective anti-inflammatory cyclooxygenase (COX-1 and COX-2) inhibitor. Inhibits COX-1 expression in HUVECS at a concentration of 5µg/ml. This COX inhibitor is indicated for the treatment of pain, fever, inflammation and stiffness. Research indicates that the S-naproxen is a more active form than the R-naproxen. The S-naproxen enantiomer was observed to inhibit platelet aggregation, and both enantiomers decrease production of thromboxane B2. Naproxen has been demonstrated to block A? fibril growth and form a complex with copper (II).
(1) W.F. Kean, et al.; J. Pharm. Sci. 78, 324 (1989) | (2) T. Zyglewska, et al.; Biochim. Biophys. Acta 1131, 78 (1992) | (3) J. Barnett, et al.; Biochim. Biophys. Acta 1209, 130 (1994) | (4) O. Laneuville, et al.; J. Pharmacol. Exp. Ther. 271, 927 (1994) | (5) N.M. Davies & K.E. Anderson; Clin. Pharmacokinet. 32, 268 (1997) | (6) N. Monck; IDrugs 6, 593 (2003) | (7) T. Takeda, et al.; J. Phys. Chem. B. 114, 15394 (2010) | (8) K.C. Duggan, et al.; J. Biol. Chem. 285, 34950 (2010)





