Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
Chemodex
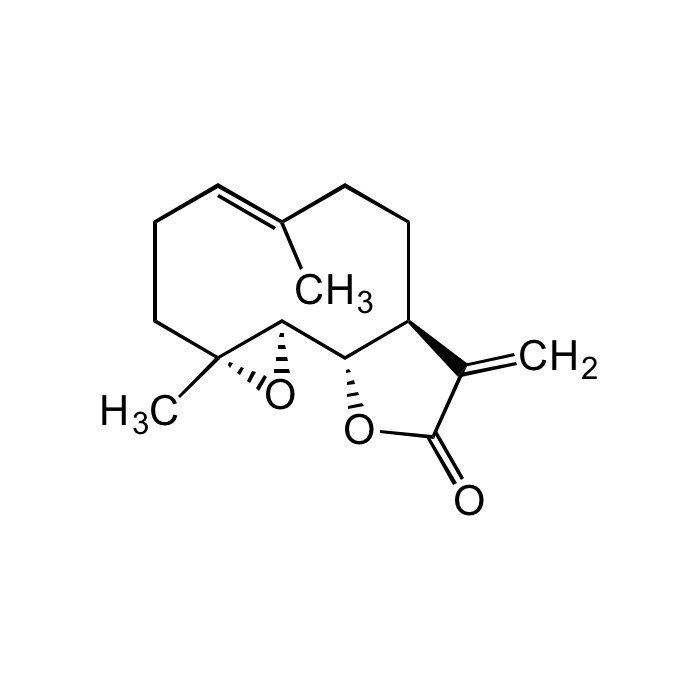
Parthenolide
| Product Details | |
|---|---|
| Synonyms | 2,3,6,7,7aS,8,10aS,10bR-octahydro-1aR,5-dimethyl-8-methylene-(4E)-oxireno[9,10]cyclodeca[1,2-b]furan-9(1aH)-one |
| Product Type | Chemical |
| Properties | |
| Formula |
C15H20O3 |
| MW | 248.32 |
| CAS | 20554-84-1 |
| Source/Host Chemicals | Synthetic. |
| Purity Chemicals | ≥98% (HPLC) |
| Appearance | White to off-white powder. |
| Solubility | Soluble in DMSO, ethanol or chloroform. |
| Identity | Determined by 1H-NMR. |
| Declaration | Manufactured by Chemodex. |
| Other Product Data |
Click here for Original Manufacturer Product Datasheet |
| InChi Key | KTEXNACQROZXEV-PVLRGYAZSA-N |
| Smiles | C/C1=C\CC[C@](O2)(C)[C@H]2[C@@H](OC(C3=C)=O)[C@H]3CC1 |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | +4°C |
| Handling Advice | Protect from light and moisture. |
| Use/Stability | Stable for at least 2 years after receipt when stored at +4°C. |
| Documents | |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Anticancer and antiangiogenic compound. Induces apoptosis, cell cycle arrest and autophagy in various cancer cell lines. At low concentrations acts as antioxidant reducing oxidative stress generated through the TCR signaling pathway. At high concentration induces O2- and causes oxidative-stress-mediated apoptosis. Potent anti-inflammatory and anti-atherosclerotic agent. NF-κB inhibitor by directly targeting IκB kinase. NLRP3 inflammasome inhibitor. Inhibits ATPase activity of NLRP3 and protease activity of caspase 1. Specifically inhibits HDAC1 without affecting other class I/II HDACs. DNA methyltransferase 1 (DNMT1) inhibitor. Reduces the expression of VEGF and its receptors VEGRF1 and 2. Microtubule-interfering compound, inhibiting cell migration and tubule formation. Anti-leishmanial and anti-trypanosomal agent.
(1) R.M. Wiedhopf, et al.; J. Pharm. Sci. 62, 345 (1973) | (2) B.H. Kwok, et al.; Chem. Biol. 8, 759 (2001) | (3) P. Pozarowski, et al.; Cell Cycle 2, 377 (2003) | (4) T.S. Tiuman, et al.; Antimicrob. Agents Chemother. 49, 176 (2005) | (5) M. Li-Weber, et al.; Cell Death Differ. 12, 408 (2005) | (6) Y.N. Gopal, et al.; Chem. Biol. 14, 813 (2007) | (7) E. Izumi, et al.; Exp. Parasitol. 118, 324 (2008) | (8) Z. Liu, et al.; J. Pharmacol. Exp. Ther. 329, 505 (2009) | (9) C. Juliana, et al.; J. Biol. Chem. 285, 9792 (2010) | (10) L. Zhao, et al.; Nutr. Rev. 69, 310 (2011) (Review) | (11) V.B. Mathema, et al.; Inflammation 35, 560 (2012) (Review) | (12) S.L. Kim, et al.; Int. J. Mol. Med. 33, 1261 (2014)






