Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
Chemodex
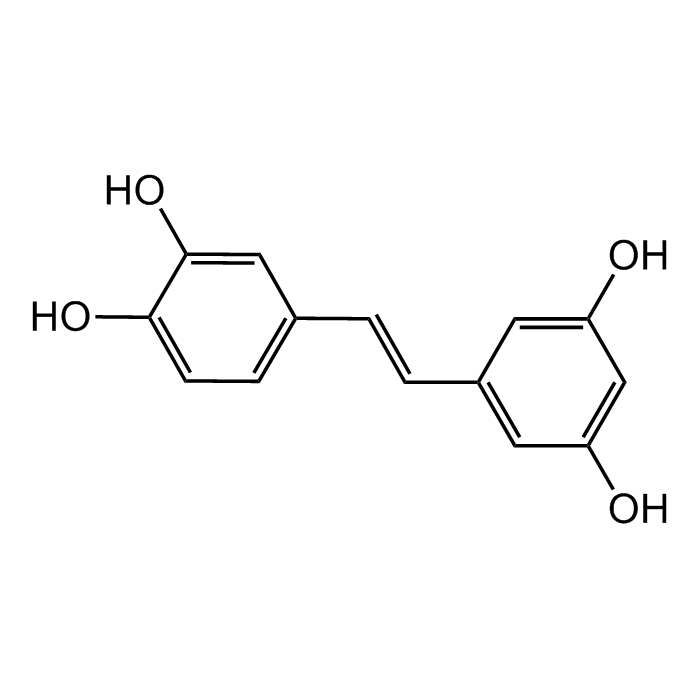
trans-Piceatannol
| Product Details | |
|---|---|
| Synonyms | 3,3',4,5'-Stilbenetetrol; 3,3',4,5'-Tetrahydroxy-trans-stilbene; Astringenin; CCRIS 9289; NSC365798; NSC622471; 3-Hydroxyresveratrol |
| Product Type | Chemical |
| Properties | |
| Formula |
C14H12O4 |
| MW | 244.24 |
| CAS | 10083-24-6 |
| Source/Host Chemicals | Plant |
| Purity Chemicals | ≥98% (HPLC) |
| Appearance | Light tan to yellow powder. |
| Solubility | Soluble in DMSO (10 mg/mL) or ethanol (10 mg/mL). |
| Identity | Determined by 1H-NMR. |
| Declaration | Manufactured by Chemodex. |
| Other Product Data |
Click here for Original Manufacturer Product Datasheet |
| InChi Key | CDRPUGZCRXZLFL-OWOJBTEDSA-N |
| Smiles | OC1=CC(/C=C/C2=CC(O)=CC(O)=C2)=CC=C1O |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +20°C |
| Long Term Storage | +4°C |
| Handling Advice | Protect from light and moisture. |
| Use/Stability | Stable for at least 2 years after receipt when stored at +4°C. |
| Documents | |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Piceatannol is a resveratrol analog with anticancer, anti-inflammatory, antioxidant, neuroprotective and cardioprotective properties. It is a spleen tyrosine kinase (Syk) inhibitor, however it exhibits antiproliferative activity by inhibiting a wide range of tyrosine and serine/threonine protein kinases, including Syk, p56lck, PKA, PKC, MLCK, CDPK, JNK and PI3K. It suppresses NF-κ activation through inhibition of IκBα kinase and NLRP3 activation. It is an apoptosis and autophagy inducer, cell cycle inhibitor, activator of SIRT1 and also inhibits adipogenesis.
(1) R.L. Geahlen, et al.; BBRC 165, 241 (1989) | (2) B.H. Wang, et al.; Planta Med. 64, 195 (1998) | (3) T. Wieder, et al.; Leukemia 15, 1735 (2001) | (4) F. Wolter, et al.; J. Nutr. 132, 298 (2002) | (5) K. Ashikawa, et al.; J. Immunol. 169, 6490 (2002) | (6) K.T. Howitz, et al.; Nature 425, 191 (2003) | (7) B.S. Wung, et al.; Pharmacol. Res. 53, 113 (2006) | (8) Z. Ovesna, et al.; Oncol. Rep. 16, 617 (2006) | (9) H.J. Kim, et al.; Ann. N Y Acad. Sci. 1095, 473 (2007) | (10) Y.H. Kim, et al.; Oncol. Rep. 19, 961 (2008) | (11) K.H. Choi, et al.; Cardiovasc. Res. 85, 836 (2010) | (12) H. Piotrowska, et al.; Mutat. Res. 750, 60 (2012) (Review) | (13) J.Y. Kwon, et al.; J. Biol. Chem. 287, 11566 (2012) | (14) Y. Kita, et al.; J. Biomed. Biotechnol. 2012, 672416 (2012) | (15) M. Minakawa, et al.; BBRC 422, 469 (2012) | 16) F. Pietrocola, et al.; Cell Cycle 11, 3851 (2012) | (17) Y.J. Surh & H.K. Na; Adv. Exp. Med. Biol. 928, 185 (2016) (Review) | (18) J. Kershaw & K.H. Kim; J. Med. Food 20, 427 (2017) (Review) | (19) J. Wen, et al.; Int. Immunopharmacol. 64, 131 (2018) | (20) K. Banik, et al.; Pharmacol. Res. 153, 104635 (2020) (Review)





