Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
Chemodex
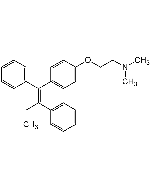
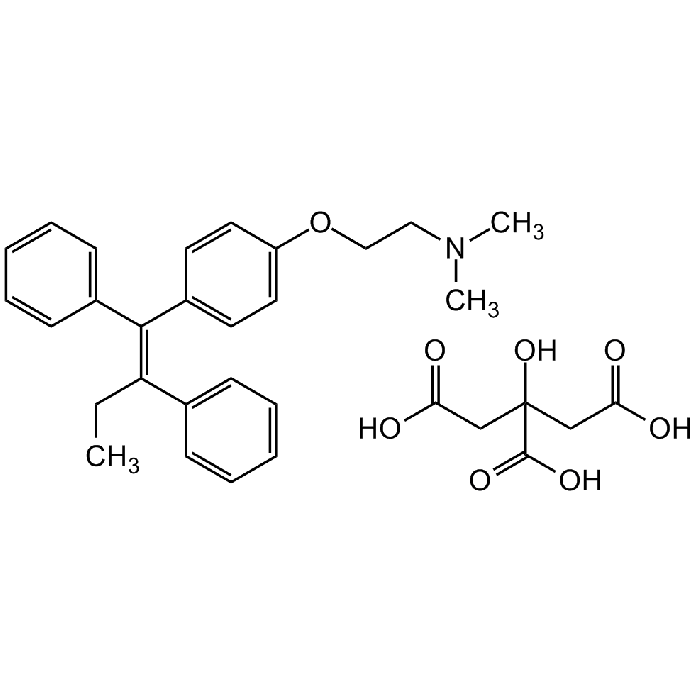
Tamoxifen citrate salt
| Product Details | |
|---|---|
| Synonyms | (Z)-1-(p-Dimethylaminoethoxyphenyl)-1,2-diphenyl-1-butene; ICI 46474; Istubal; Kessar; Noltam; Nolvadex; TMX; Valodex; Zemide |
| Product Type | Chemical |
| Properties | |
| Formula | C26H29NO . C6H8O7 |
| MW | 563.64 |
| CAS | 54965-24-1 |
| Purity Chemicals | ≥98% |
| Appearance | White powder. |
| Solubility | Soluble in DMSO or methanol. Slightly soluble in ethanol. |
| Identity | Determined by NMR. |
| Declaration | Manufactured by Chemodex. |
| Other Product Data |
Click here for Original Manufacturer Product Datasheet |
| InChi Key | FQZYTYWMLGAPFJ-OQKDUQJOSA-N |
| Smiles | CN(CCOC1=CC=C(/C(C2=CC=CC=C2)=C(C3=CC=CC=C3)/CC)C=C1)C.OC(CC(O)(C(O)=O)CC(O)=O)=O |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +20°C |
| Long Term Storage | +4°C |
| Handling Advice |
Keep cool and dry. Protect from light and moisture. |
| Use/Stability | Stable for at least 2 years after receipt when stored at +4°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Selective estrogen receptor modulator (SERM) used as adjuvant therapy for estrogen-dependent breast cancer. Antagonist of ER action in breast tissue and breast cancer cells and ER agonist in bone, uterus, and the cardiovasculature system. This prodrug is metabolized by cytochrome P450 (CYP450) enzymes to the active metabolites N-desmethyl-TMX, 4-hydroxy-N-desmethyl-TMX (endoxifen) and 4-hydroxy-TMX (afimoxifene). N,N-Didesmethyl-4-hydroxytamoxifen (norendoxifen), another active metabolite has been found to act as a potent competitive aromatase inhibitor and may also be involved in its antiestrogenic activity. Binds microsomal antiestrogen binding sites, altering cholesterol esterification at therapeutic doses and impacting breast cancer cell differentiation, apoptosis, and autophagy. Protein kinase C (PKC) inhibitor and anti-angiogenetic factor.
(1) K. B. Horwitz & W. L. McGuire; J. Biol. Chem. 253, 8185 (1978) | (2) V.C. Jordan, et al.; Cancer Treat. Rep. 64, 745 (1980) | (3) C.A. O'Brian, et al.; Cancer Res. 45, 2462 (1985) | (4) K.J. Edwards, et al.; J. Med. Chem. 35, 2753 (1992) | (5) M. Clarke, et al.; Lancet 351, 1451 (1998) | (6) D.A. Tonetti & V.C. Jordan; Mol. Med. Today 2, 218 (1996) | (7) J.M. Hall, et al.; J. Biol. Chem. 276, 36869 (2001) | (8) P. Thomas, et al.; Endocrinol. 146, 624 (2005) | (9) M.S. Singh, et al.; Breast 20, 111 (2011) | (10) P. de Medina, et al.; Chem. Phys. Lip. 164, 432 (2011) | (11) V.K. Todorova, et al.; Cancer Chemother. Pharmacol. 67, 285 (2011)