Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
Chemodex
Tetrakis(trimethylsilyl)silane
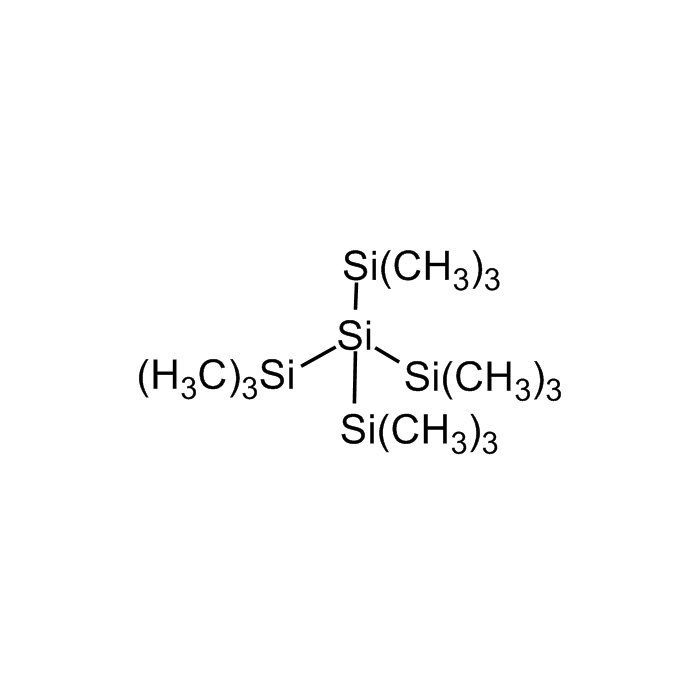
| Product Details | |
|---|---|
| Synonyms | 2,2-Bis(trimethylsilyl)-1,1,1,3,3,3-hexamethyl-trisilane; TTMSS |
| Product Type | Chemical |
| Properties | |
| Formula | (CH3)3Si]4Si |
| MW | 320.84 |
| CAS | 4098-98-0 |
| Source/Host Chemicals | Synthetic |
| Purity Chemicals | ≥97% (GC) |
| Appearance | White to off-white powder. |
| Solubility | Soluble in chloroform (100mg/ml). |
| Identity | Determined by 1H-NMR. |
| Declaration | Manufactured by Chemodex. |
| Other Product Data |
Click here for Original Manufacturer Product Datasheet |
| InChi Key | BOJSDHZZKKYWAS-UHFFFAOYSA-N |
| Smiles | C[Si](C)(C)[Si]([Si](C)(C)C)([Si](C)(C)C)[Si](C)(C)C |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | +4°C |
| Handling Advice | Protect from light and moisture. |
| Use/Stability | Stable for at least 2 years after receipt when stored at +4°C. |
| Documents | |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Tetrakis(trimethylsilyl)silane (TTMSS) is an organosilicon compound. TTMSS is widely used as a reagent and precursor in organic synthesis and silicon-based chemistry. TTMSS is commonly used as a protecting group for various functional groups in organic synthesis, in deoxygenation reactions to remove oxygen-containing functional groups like ketones, aldehydes, and esters, converting them into the corresponding trimethylsilyl ethers. TTMSS is utilized in silylation reactions to introduce trimethylsilyl groups onto organic compounds, enhancing their volatility and stability for gas chromatography and mass spectrometry analysis. Due to its diverse applications in organic chemistry, Tetrakis(trimethylsilyl)silane has become a valuable tool for synthetic chemists, particularly in the fields of silicon-based chemistry, protecting group strategies, and cross-coupling reactions.
(1) C.H. Ming, et al.; J. Organomet. Chem. 316, 255 (1986) | (2) J. Lalevee, et al.; J. Org. Chem. 72, 6434 (2007) | (3) E. Perret, et al.; J. Phys. Condens. Matter 22, 235102 (2010) | (4) L.D. Funt, et al.; J. Org. Chem. 82, 7583 (2017) | (5) C. Chatgilialoglu, et al.; Chem. Rev. 118, 6516 (2018) | (6) Y. Lee, et al.; J. Nanosci. Nanotechnol. 21, 2139 (2021) | (7) S. George, et al.; Org. Lett. 25, 4345 (2023)





