Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
AdipoGen Life Sciences
anti-NLRP1b (mouse), mAb (2A12)
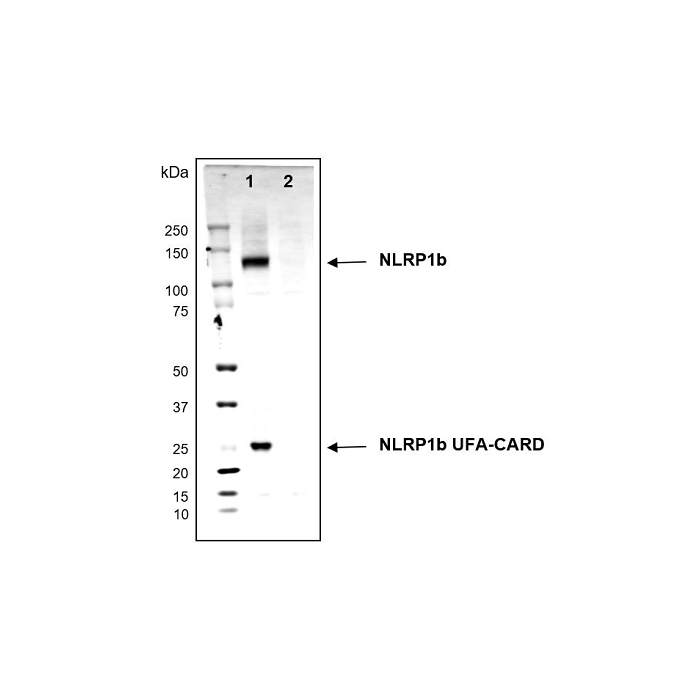
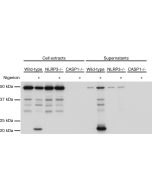
Method: Cell extracts (about 30µg) from the mouse cells Raw264.7 (lane 1) or Raw264.7 NLRP1b KO (lane 2) are separated by SDS-PAGE under reducing conditions, transferred to nitrocellulose and incubated with the NLRP1b, mAb (2A12) (1µg/ml). After incubation with anti-mouse Alexa 633 conjugated antibody, proteins are visualized by a fluorescence detection system at 700nm. Picture Courtesy of Dr Andrew Sandstrom, UT Southwestern Medical Center.
| Product Details | |
|---|---|
| Synonyms | NACHT, LRR and PYD domains-containing protein 1b allele 1; Nalp1b |
| Product Type | Monoclonal Antibody |
| Properties | |
| Clone | 2A12 |
| Isotype | Mouse IgG2b |
| Source/Host | Purified from concentrated hybridoma tissue culture supernatant. |
| Immunogen/Antigen | CARD domain of mouse NLRP1b. |
| Application |
Western Blot: (1:1’000) |
| Crossreactivity |
Mouse Rat |
| Specificity |
Recognizes mouse and rat NLRP1b. Detects mouse NLRP1b and also both the B6 and 129 alleles of murine NLRP1b. Does not cross-react with human NLRP1. |
| Purity | ≥95% (SDS-PAGE) |
| Concentration | 1 mg/ml |
| Formulation | Liquid. In PBS containing 10% glycerol and 0.02% Proclin 300. |
| Isotype Negative Control | |
| Accession Number | Q2LKW6 |
| Shipping and Handling | |
| Shipping | BLUE ICE |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice |
After opening, prepare aliquots and store at -20°C. Avoid freeze/thaw cycles. |
| Use/Stability | Stable for at least 1 year after receipt when stored at -20°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
The NLRP1 inflammasome is a multiprotein complex that is a potent activator of inflammation. As inflammasome-forming sensor protein, NLRP1b, upon detection of microbial molecules or pathogen-encoded activities, serves as a platform for the recruitment and activation of proinflammatory caspases including caspase-1 through a caspase activation and recruitment domain (CARD). Active caspase-1 mediates the maturation and release of the proinflammatory cytokines interleukin IL-1β and IL-18.
Mouse NLRP1b can be activated through proteolytic cleavage by the bacterial Lethal Toxin (LeTx) protease, resulting in degradation of the N-terminal domains of NLRP1b and liberation of the bioactive C-terminal domain, which includes the caspase activation and recruitment domain (CARD). NLRP1b has an unusual domain architecture, containing a CARD at its C-terminus rather than the N-terminus like all other inflammasomes, and a function-to-find domain (FIIND), which is located between the LRRs and CARD. The FIIND undergoes a constitutive self-cleavage event, such that NLRP1b exists in its non-activated state as two, noncovalently associated polypeptides, the N-terminal domains and the C-terminal CARD-containing fragment. Inflammasome activation is the result of site-specific cleavage in the N-terminus of mouse NLRP1b by the Lethal Factor (LF) protease subunit of LeTx, which results in its activation. Upon cleavage by LF, a novel N-terminus is formed, which is then targeted for proteasomal degradation by a protein quality control mechanism called the ‘N-end rule’ pathway. Since the proteasome is a processive protease, it progressively degrades the N-terminal domains of NLRP1b but is disengaged upon arriving at the self-cleavage site within the FIIND domain. Degradation of the N-terminal domains thus releases the bioactive C-terminal CARD-containing fragment of NLRP1b, which is sufficient to initiate downstream inflammasome activation.
Genetic variations in the NLRP1/NALP1 gene are associated with susceptibility to vitiligo-associated multiple autoimmune disease type 1, an autoimmune disorder characterized by the association of vitiligo with several autoimmune and autoinflammatory diseases including autoimmune thyroid disease, rheumatoid arthritis and systemic lupus erythematosus.
- Functional degradation: a mechanism of NLRP1 inflammasome activation by diverse pathogen enzymes: A. Sandstrom, et al.; Science 364, eaau1330 (2019)
- N-terminal degradation activates the NLRP1B inflammasome: A.J. Chui, et al.; Science 364, 82 (2019)