Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
AdipoGen Life Sciences
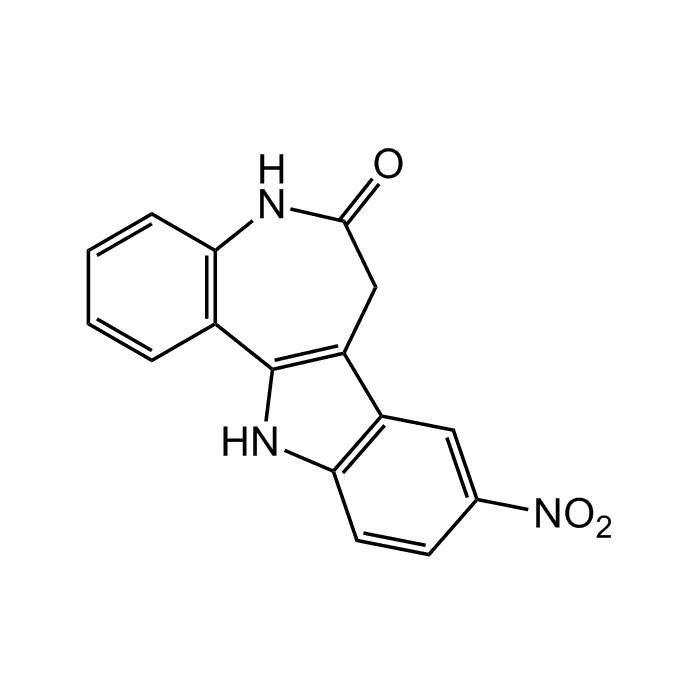
Alsterpaullone
As low as
50
CHF
CHF 50.00
In stock
Only %1 left
AG-CR1-0036-M0011 mgCHF 50.00
AG-CR1-0036-M0055 mgCHF 150.00
| Product Details | |
|---|---|
| Synonyms | 9-Nitro-7,12-dihydroindolo-[3,2-d][1]benzazepin-6(5H)-one |
| Product Type | Chemical |
| Properties | |
| Formula |
C16H11N3O3 |
| MW | 293.3 |
| CAS | 237430-03-4 |
| Purity Chemicals | ≥98% (NMR) |
| Appearance | Yellow to brown powder. |
| Solubility | Soluble in DMSO; insoluble in water or 100% ethanol. |
| Identity | Determined by IR, 1H-NMR and 13C-NMR. |
| InChi Key | OLUKILHGKRVDCT-UHFFFAOYSA-N |
| Smiles | [O-][N+](=O)C1=CC2=C(NC3=C2CC(=O)NC2=C3C=CC=C2)C=C1 |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | +4°C |
| Handling Advice | Protect from light. |
| Use/Stability | Stable for at least 2 years after receipt when stored at +4°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
- Potent CDK1/cyclin B (IC50 = 35 nM) inhibitor [1].
- Anti-tumor compound [1]
- Potent inhibitor of CDK2/cyclin A, CDK2/cyclin E (IC50 = 200 nM), CDK5/p25 (IC50 = 40 nM), CDK5/p35 (IC50 = 40 nM) [2, 4].
- GSK-3β (glycogen synthase kinase-3β) inhibitor [2, 3].
- Apoptosis inhibitor [6, 7].
- Apoptosis inducer [5].
- Angiogenesis inhibitor [8].
Product References
- Paullones, a series of cyclin-dependent kinase inhibitors: synthesis, evaluation of CDK1/cyclin B inhibition, and in vitro antitumor activity: C. Schultz, et al.; J. Med. Chem. 42, 2909 (1999)
- Paullones are potent inhibitors of glycogen synthase kinase-3beta and cyclin-dependent kinase 5/p25: M. Leost, et al.; Eur. J. Biochem. 267, 5983 (2000)
- Intracellular Targets of Paullones. Identification following affinity purification on immobilized inhibitor: M. Knockaert, et al.; J. Biol. Chem. 277, 25493 (2002)
- The specificities of protein kinase inhibitors: an update: J. Bain, et al.; Biochem. J. 371, 199 (2003)
- Alsterpaullone, a novel cyclin-dependent kinase inhibitor, induces apoptosis by activation of caspase-9 due to perturbation in mitochondrial membrane potential: T. Lahusen, et al.; Mol Carcinog 36, 183 (2003)
- Thapsigargin-induced apoptosis was prevented by glycogen synthase kinase-3 inhibitors in PC12 cells: T. Takadera, et al.; Neurosci. Lett. 408, 124
- Colchicine-induced apoptosis was prevented by glycogen synthase kinase-3 inhibitors in PC12 cells: T. Takadera, et al.; Cell. Mol. Neurobiol. 30, 863 (2010)
- Anti-angiogenic potential of small molecular inhibitors of cyclin dependent kinases in vitro: S. Zahler, et al.; Angiogenesis 13, 239 (2010)