Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
AdipoGen Life Sciences
LY-294,002
As low as
20
CHF
CHF 20.00
In stock
Only %1 left
AG-CR1-0108-M0011 mgCHF 20.00
AG-CR1-0108-M0055 mgCHF 40.00
AG-CR1-0108-M02525 mgCHF 110.00
| Product Details | |
|---|---|
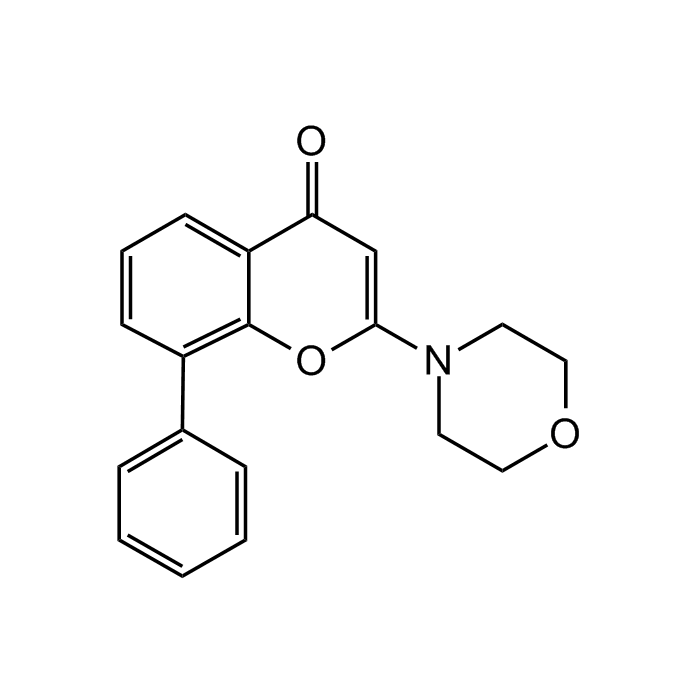
| Synonyms | 2-(4-Morpholinyl)-8-phenyl-4H-1-benzopyran-4-one |
| Product Type | Chemical |
| Properties | |
| Formula |
C19H17NO3 |
| MW | 307.4 |
| CAS | 154447-36-6 |
| Purity Chemicals | ≥99% (NMR) |
| Appearance | White to off-white crystalline solid. |
| Solubility | Soluble in DMSO, 100% ethanol or methanol. |
| Identity | Determined by 1H-NMR. |
| InChi Key | CZQHHVNHHHRRDU-UHFFFAOYSA-N |
| Smiles | O=C1C=C(OC2=C(C=CC=C12)C1=CC=CC=C1)N1CCOCC1 |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Use/Stability | Stable for at least 2 years after receipt when stored at -20°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
- Potent, cell permeable, highly specific, reversible PI(3)K (phosphoinositide 3-kinase) inhibitor.
- pan-class I / II / III PI3K inhibitor.
- Acts on the ATP-binding site of the enzyme.
- In solution more stable than wortmannin.
- Antagonizes P-glycoprotein-mediated multidrug resistance.
- Blocks Akt phosphorylation.
- Pim-1 kinase inhibitor.
- Autophagy inhibitor (autophagosome formation).
- Inhibitor of BET bromodomain proteins BRD2, BRD3 and BRD4.
- Angiogenesis inhibitor.
Product References
- A specific inhibitor of phosphatidylinositol 3-kinase, 2-(4- morpholinyl)-8-phenyl-4H-1-benzopyran-4-one (LY294002): C.J. Vlahos, et al.; J. Biol. Chem. 269, 5241 (1994)
- Investigation of neutrophil signal transduction using a specific inhibitor of phosphatidylinositol 3-kinase: C.J. Vlahos, et al.; J. Immunol. 154, 2413 (1995)
- Pim-1 Ligand-bound Structures Reveal the Mechanism of Serine/Threonine Kinase Inhibition by LY294002: M.D. Jacobs, et al.; J. Biol. Chem. 280, 13728 (2005)
- LY294002 and LY303511 Sensitize Tumor Cells to Drug-Induced Apoptosis via Intracellular Hydrogen Peroxide Production Independent of the Phosphoinositide 3-Kinase-Akt Pathway: T.W. Poh & S. Pervaiz; Cancer Res. 65, 6264 (2005)
- LY294,002, a specific inhibitor of PI3K/Akt kinase pathway, antagonizes P-glycoprotein-mediated multidrug resistance: M. Barancík, et al.; Eur. J. Pharm. Sci. 29, 426 (2006)
- A role for Akt in the rapid regulation of inflammatory and apoptotic pathways in mouse bladder: F.J. Tamarkin, et al.; Naunyn Schmiedebergs Arch. Pharmacol. 373, 349 (2006)
- Synthesis and biological evaluation of sulfonylhydrazone-substituted imidazo[1,2-a]pyridines as novel PI3 kinase p110alpha inhibitors: M. Hayakawa, et al.; Bioorg. Med. Chem. 15, 5837 (2007)
- The commonly used PI3-kinase probe LY294002 is an inhibitor of BET bromodomains: A. Dittmann, et al.; ACS Chem. Biol. 9, 495 (2014)






