Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
AdipoGen Life Sciences
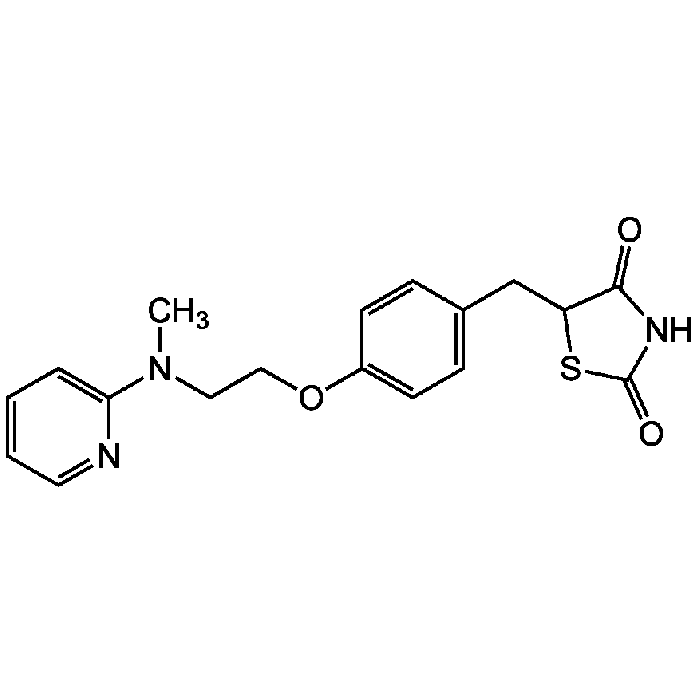
Rosiglitazone
As low as
40
CHF
CHF 40.00
In stock
Only %1 left
AG-CR1-3570-M01010 mgCHF 40.00
AG-CR1-3570-M02525 mgCHF 80.00
AG-CR1-3570-M100100 mgCHF 200.00
AG-CR1-3570-G0011 gCHF 490.00
| Product Details | |
|---|---|
| Synonyms | BRL 49653; HSDB 7555; TDZ 01 |
| Product Type | Chemical |
| Properties | |
| Formula |
C18H19N3O3S |
| MW | 357.4 |
| Merck Index | 14: 8265 |
| CAS | 122320-73-4 |
| Purity Chemicals | ≥98% (HPLC) |
| Appearance | White to off-white powder. |
| Solubility | Soluble in DMSO, ethanol or methanol. |
| Identity | Determined by 1H-NMR. |
| InChi Key | YASAKCUCGLMORW-UHFFFAOYSA-N |
| Smiles | CN(CCOC1=CC=C(CC2SC(=O)NC2=O)C=C1)C1=CC=CC=N1 |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | +4°C |
| Use/Stability |
Stable for at least 2 years after receipt when stored at +4°C. Stock solutions are stable for at least 3 months when stored at -20°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
- Antidiabetic, hypoglycemic agent [1, 5].
- Potent and selective peroxisome proliferator-activated receptor γ (PPAR-γ) agonist [2, 3].
- Potent insulin sensitizing agent binding to the PPAR receptors in fat cells and making the cells more responsive to insulin. Ameliorates insulin resistance [4-6, 8].
- Improves blood pressure and vascular function [7].
- Enhances proliferation of endogenous neural progenitor cells (NPCs) [9].
- Anti-inflammatory compound [10, 11].
- Has controversial therapeutic effects on the cardiovascular system [12].
- Promotes adipocyte differentiation of mesenchymal stem cells (MSCs) [13].
Product References
- [[ω-(Heterocyclylamino)alkoxy]benzyl]-2,4-thiazolidinediones as potent antihyperglycemic agents: B.C. Cantello, et al.; J. Med. Chem. 37, 3977 (1994)
- An antidiabetic thiazolidinedione is a high affinity ligand for peroxisome proliferator-activated receptor γ (PPARγ): J.M. Lehmann, et al.; J. Biol. Chem. 270, 12953 (1995)
- Rosiglitazone: J.A. Balfour & G.L. Plosker; Drugs 57, 921 (1999) (Review)
- Differential effects of insulin-sensitizers troglitazone and rosiglitazone on ion currents in rat vascular myocytes: G.A. Knock, et al.; Eur. J. Pharmacol. 368, 103 (1999)
- Rosiglitazone: an agent from the thiazolidinedione class for the treatment of type 2 diabetes: A. Cheng-Lai & A. Levine; Heart Dis. 2, 326 (2000) (Review)
- Rosiglitazone in the treatment of type 2 diabetes mellitus: a critical review: J.M. Malinowski & S. Bolesta; Clin. Ther. 22, 1151 (2000) (Review)
- PPAR(gamma) agonist rosiglitazone improves vascular function and lowers blood pressure in hypertensive transgenic mice: M.J. Ryan, et al.; Hypertension 3, 661 (2004)
- Rosiglitazone: a review of its use in type 2 diabetes mellitus: E.D. Deeks & S.J. Keam; Drugs 67, 2747 (2007) (Review)
- Rosiglitazone enhances the proliferation of neural progenitor cells and inhibits inflammation response after spinal cord injury: Q.Q. Meng, et al.; Neurosci. Lett. 503, 191 (2011)
- Inhibitory effects of rosiglitazone on lipopolysaccharide-induced inflammation in a murine model and HK-2 cells: W.M. Wang, et al.; Am. J. Nephrol. 34, 152 (2011)
- PPARγ agonist rosiglitazone ameliorates LPS-induced inflammation in vascular smooth muscle cells via the TLR4/TRIF/IRF3/IP-10 signaling pathway: Y. Ji, et al.; Cytokine 55, 409 (2011)
- PPARγ activator, rosiglitazone: Is it beneficial or harmful to the cardiovascular system? S. Palee, et al.; World J. Cardiol. 3, 144 (2011)
- Rosiglitazone-induced adipogenesis in a bone marrow mesenchymal stem cell line: D. Wang, et al.; Biomed. Sci. Instrum. 47, 213 (2011)
- ROCK2 inhibition enhances the thermogenic program in white and brown fat tissue in mice: L. Wei, et al.; FASEB J. ahead of print (2019)