Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
AdipoGen Life Sciences
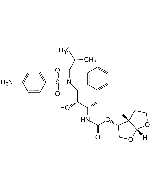
Ritonavir
As low as
50
CHF
CHF 50.00
In stock
Only %1 left
AG-CR1-3683-M01010 mgCHF 50.00
AG-CR1-3683-M05050 mgCHF 140.00
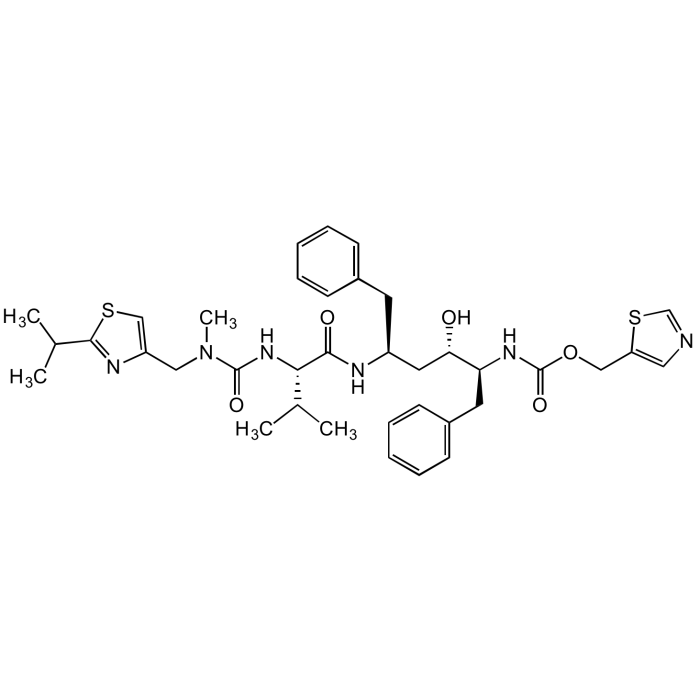
| Product Details | |
|---|---|
| Synonyms | RTV; A-84538; ABT-538; Abbot 84538; NSC 693184 |
| Product Type | Chemical |
| Properties | |
| Formula |
C37H48N6O5S2 |
| MW | 720.9 |
| CAS | 155213-67-5 |
| RTECS | XA5310000 |
| Purity Chemicals | ≥98% (HPLC) |
| Appearance | White to off-white solid. |
| Solubility | Soluble in DMSO (10mg/ml), DMF (10mg/ml) or EtOH. |
| Other Product Data |
Optical Rotation [α]D20 (c=2, MeOH): +7.0° to +10.0°. |
| InChi Key | NCDNCNXCDXHOMX-XGKFQTDJSA-N |
| Smiles | CC(C1=NC(CN(C(N[C@H](C(N[C@H](C[C@@H]([C@@H](NC(OCC2=CN=CS2)=O)CC3=CC=CC=C3)O)CC4=CC=CC=C4)=O)C(C)C)=O)C)=CS1)C |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice | Keep cool and dry. |
| Use/Stability | Stable for at least 2 years after receipt when stored at -20°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
- HIV-1 and HIV-2 protease inhibitor.
- Glucose transporter 1 (GLUT1) and 4 (GLUT4) inhibitor. Inhibits transport of glucose across the plasma membranes of mammalian cells and consequently decreases glycolysis.
- Useful agent for immunometabolism research.
- Antitumor agent. Apoptosis inducer and cell proliferation inhibitor in GLUT1/GLUT4 overexpressed cancer cell lines.
- Tumor cells rely on elevated glucose consumption and metabolism for survival and proliferation. Glucose transporters mediating glucose entry are key proximal rate-limiting checkpoints. Among the fourteen SLC2A family members, GLUTs 1 and 4 are high-affinity glucose transporters.
- Inhibits 20S proteasome chymotrypsin-like activity.
- The proteinase inhibitor Kaletra, which is a mixture of the HIV-1 proteinase inhibitors lopinavir and ritonavir has been shown to be effective against SARS-CoV and MERS-CoV.
- Shown in a SARS-CoV-2 protease structure model study to potentially bind and inhibit the coronavirus endopeptidase C30 (CEP_C30) of SARS-CoV-2. An initial randomized trial study was not successful.
Product References
- ABT-538 is a potent inhibitor of human immunodeficiency virus protease and has high oral bioavailability in humans: D.J. Kempf, et al.; PNAS 92, 2484 (1995)
- Discovery of ritonavir, a potent inhibitor of HIV protease with high oral bioavailability and clinical efficacy: D.J. Kempf, et al.; J. Med. Chem. 41, 602 (1998)
- How an Inhibitor of the HIV-I Protease Modulates Proteasome Activity: G. Schmidtke, et al.; J. Biol. Chem. 274, 35734 (1999)
- Pharmacological and therapeutic properties of ritonavir-boosted protease inhibitor therapy in HIV-infected patients: R.K. Zeldin, et al.; J. Antimicrob. Chemother. 53, 4 (2004)
- Molecular dynamic simulations analysis of ritonavir and lopinavir as SARS-CoV 3CL(pro) inhibitors: V. Nukoolkarn, et al.; J. Theor. Biol. 254, 861 (2008)
- HIV protease inhibitors act as competitive inhibitors of the cytoplasmic glucose binding site of GLUTs with differing affinities for GLUT1 and GLUT4: R.C. Hresko & P.W. Hruz; PLoS One 6, e25237 (2011)
- Targeting Glycolysis and Compensatory Mitochondrial Metabolism In An In Vivo Xenograft Model Of Multiple Myeloma With FDA Approved Ritonavir and Metformin: S. Dalva-Aydemir, et al.; Blood 122, 1922 (2013)
- Glucose transporter 1-mediated glucose uptake is limiting for B-cell acute lymphoblastic leukemia anabolic metabolism and resistance to apoptosis: T. Liu, et al.; Cell Death Dis. 5, e1470 (2014)
- In Silico Modeling-based Identification of Glucose Transporter 4 (GLUT4)-selective Inhibitors for Cancer Therapy: R.K. Mishra, et al.; J. Biol. Chem. 290, 14441 (2015)
- A guide to immunometabolism for immunologists: L.A. O'Neill, et al.; Nat. Rev. Immunol. 16, 553 (2016)
- Development of GLUT4-selective antagonists for multiple myeloma therapy: C. Wei, et al.; Eur. J. Med. Chem. 139, 573 (2017)
- Molecular Modeling Evaluation of the Binding Effect of Ritonavir, Lopinavir and Darunavir to Severe Acute Respiratory Syndrome Coronavirus 2 Proteases: S. Lin, et al.; (Epub ahead of print) (2020)
- A Trial of Lopinavir-Ritonavir in Adults Hospitalized with Severe Covid-19: B. Cao, et al.; N. Engl. J. Med. (Epub ahead of print) (2020)