Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
AdipoGen Life Sciences
U-73122
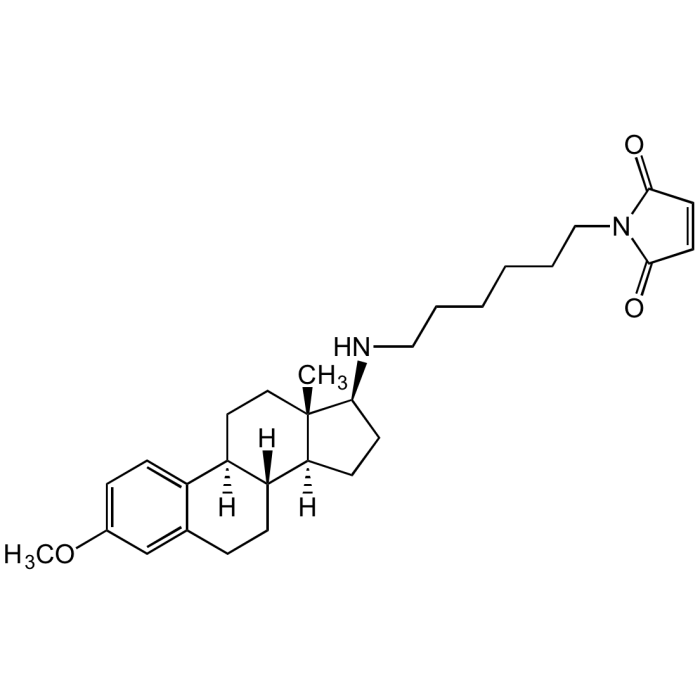
| Product Details | |
|---|---|
| Synonyms | 1-[6-[[(17β)-3-Methoxyestra-1,3,5(10)-trien-17-yl]amino]hexyl]-1H-pyrrole-2,5-dione |
| Product Type | Chemical |
| Properties | |
| Formula |
C29H40N2O3 |
| MW | 464.6 |
| CAS | 112648-68-7 |
| RTECS | UX9626600 |
| Purity Chemicals | ≥95% |
| Appearance | White to off-white solid. |
| Solubility | Soluble in methylene chloride or chloroform (10mg/ml). Slightly soluble in DMSO (2mg/ml), DMF (2mg/ml) or 100% ethanol (1mg/ml). Sparingly soluble in aqueous solutions. See also Solubility Note below. |
| Other Product Data |
Solubility/Stability and Application NOTE: U-73122 is only sparingly soluble in aqueous media. Therefore the compound is delivered to cells either after dissolving in organic solvents or complexing with serum proteins. Preparation of a Working solution: Stability: Application: |
| InChi Key | LUFAORPFSVMJIW-ZRJUGLEFSA-N |
| Smiles | C[C@@]12[C@](CC[C@@H]2NCCCCCCN3C(C=CC3=O)=O)([H])[C@]4([H])CCC5=CC(OC)=CC=C5[C@@]4([H])CC1 |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice |
Keep cool and dry. Protect from light. Protect from moisture. |
| Use/Stability | Stable for at least 2 years after receipt when stored at -20°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
- Gasdermin D N-terminal fragment (GSDMD-N)-induced pyroptosis inhibitor. Protects against GSDMD-N cytotoxicity in macrophages or against lethal infection in mice.
- Useful agent for inflammasome research.
- Potent cell permeable phospholipase C (PLC) , phospholipase A2 and 5-LO (5-lipoxygenase) inhibitor. Affects the phospholipases by inhibiting the hydrolysis of PPI (phosphatidylinositol) to IP3 (inositol triphosphate), which in turn leads to a drop in cytosolic Ca2+.
- Potent inhibitor of the SERCA pump in smooth muscle. Potently inhibits Ca2+-release from isolated smooth muscle cells independent of its effect on PLC.
- Inhibits agonist-induced platelet aggregation. Potently inhibits human polymorphonuclear neutrophil adhesion on biological surfaces and exhibits antinociceptive activity in vivo. Shown to inhibit agonist-induced down-regulation of muscarinic receptors in SK-N-SH neuroblastoma cells.
- Activates TRPM4 and inhibits TRPM3 channels.
- Potent human telomerase inhibitor.
- Selective inhibition of receptor-coupled phospholipase C-dependent processes in human platelets and polymorphonuclear neutrophils: J.E. Bleasdale, et al.; J. Pharmacol. Exp. Ther. 255, 756 (1990)
- Receptor-coupled signal transduction in human polymorphonuclear neutrophils: effects of a novel inhibitor of phospholipase C-dependent processes on cell responsiveness: R.J. Smith, et al.; J. Pharmacol. Exp. Ther. 253, 688 (1990)
- The aminosteroid U-73122 inhibits muscarinic receptor sequestration and phosphoinositide hydrolysis in SK-N-SH neuroblastoma cells. A role for Gp in receptor compartmentation: A.K. Thompson, et al.; J. Biol. Chem. 266, 23856 (1991)
- Use of U-73122 as an Inhibitor of Phospholipase C-Dependent Processes: J.E. Bleasdale & S.K. Fisher; Neuroprotocols 3, 125 (1993)
- Calcium homeostasis in mouse fibroblast cells: affected by U-73122, a putative phospholipase C beta blocker, via multiple mechanisms: J.P. Grierson & J. Meldolesi; Br. J. Pharmacol. 115, 11 (1995)
- U-73122: a potent inhibitor of human polymorphonuclear neutrophil adhesion on biological surfaces and adhesion-related effector functions: R.J. Smith, et al.; J. Pharmacol. Exp. Ther. 278, 320 (1996)
- The aminosteroid phospholipase C antagonist U-73122 (1-[6-[[17-beta-3-methoxyestra-1,3,5(10)-trien-17-yl]amino]hexyl]-1H-pyrro le-2,5-dione) potently inhibits human 5-lipoxygenase in vivo and in vitro: C. Feisst, et al.; Mol. Pharmacol. 67, 1751 (2005)
- Potent inhibition of human telomerase by U-73122: Y.J. Chen, et al.; J. Biomed. Sci. 13, 667 (2006)
- The PI-PLC inhibitor U-73122 is a potent inhibitor of the SERCA pump in smooth muscle: M.A. Hollywood, et al.; Br. J. Pharmacol. 160, 1293 (2010)
- VLDL-activated cell signaling pathways that stimulate adrenal cell aldosterone production: Y.Y. Tsai, et al.; Mol. Cell. Endocrinol. 433, 138 (2016)
- Direct modulation of TRPM4 and TRPM3 channels by the phospholipase C inhibitor U73122: M.G. Leitner, et al.; Br. J. Pharmacol. 173, 2555 (2016)
- Lipid peroxidation drives Gasdermin D-mediated pyroptosis in lethal polymicrobial Sepsis: R. Kang, et al.; Cell Host Microbe 24, 97 (2018)






