Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
BioViotica
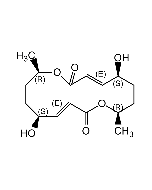
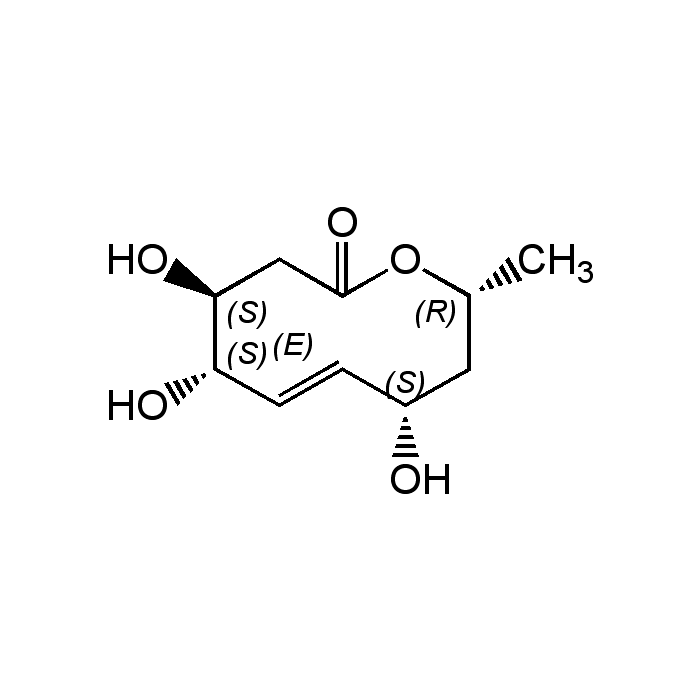
Decarestrictine D
As low as
110
CHF
CHF 110.00
In stock
Only %1 left
BVT-0283-M0011 mgCHF 110.00
| Product Details | |
|---|---|
| Synonyms | Tuckolide; (4S,5S,8S,10R,E)-4,5,8-Trihydroxy-10-methyl-3,4,5,8,9,10-hexahydro-2H-oxecin-2-one |
| Product Type | Chemical |
| Properties | |
| Formula |
C10H16O5 |
| MW | 216.2 |
| CAS | 127393-89-9 |
| Source/Host Chemicals | Isolated from Penicillium sp. |
| Purity Chemicals | ≥98% (HPLC) |
| Appearance | White to off-white solid. |
| Solubility | Soluble in DMSO, methanol or acetone. |
| Identity | Determined by 1H-NMR. |
| Declaration | Manufactured by BioViotica. |
| InChi Key | HWMMWMJBUOCCFZ-XYEXOTNWSA-N |
| Smiles | C[C@@H]1C[C@H](O)\C=C\[C@H](O)[C@@H](O)CC(=O)O1 |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Use/Stability |
Stable for at least 1 year after receipt when stored at -20°C. After reconstitution protect from light at -20°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
- Hypolipidemic.
- Cholesterol biosynthesis inhibitor.
Product References
- Chemical investigation of the metabolites from the Canadian tuckahoe, Polyporustuberaster: W.A. Ayer, et al.; J. Nat. Prod. 55, 649 (1992)
- Secondary metabolites by chemical screening. 8. Decarestrictines, a new family of inhibitors of cholesterol biosynthesis from Penicillium. I. Strain description, fermentation, isolation and properties: S. Grabley, et al.; J. Antibiot. (Tokyo) 45, 56 (1992)
- Secondary metabolites by chemical screening. 9. Decarestrictines, a new family of inhibitors of cholesterol biosynthesis from Penicillium. II. Structure elucidation of the decarestrictines A to D: A. Göhrt, et al.; J. Antibiot. (Tokyo) 45, 66 (1992)
- A non-enzymatic reaction in the late biosynthesis of the decarestrictine family: M. Mayer & R. Thiericke; J. Antibiot. (Tokyo) 46, 1372 (1993)
- Synthesis of Tuckolide, a New Cholesterol Biosynthesis Inhibitor: M.B. Andrus & T.L. Shih; J. Org. Chem. 61, 8780 (1996)
- An efficient total synthesis of decarestrictine D: P. Gupta, et al.; Eur. J. Org. Chem. 2008, 1195 (2008)
- Asymmetric synthesis of allylsilanes by the borylation of lithiated carbamates: formal total synthesis of (-)-decarestrictine D: M. Binanzer, et al.; Angew. Chem. Int. Ed. 49, 4264 (2010)