Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
BioViotica
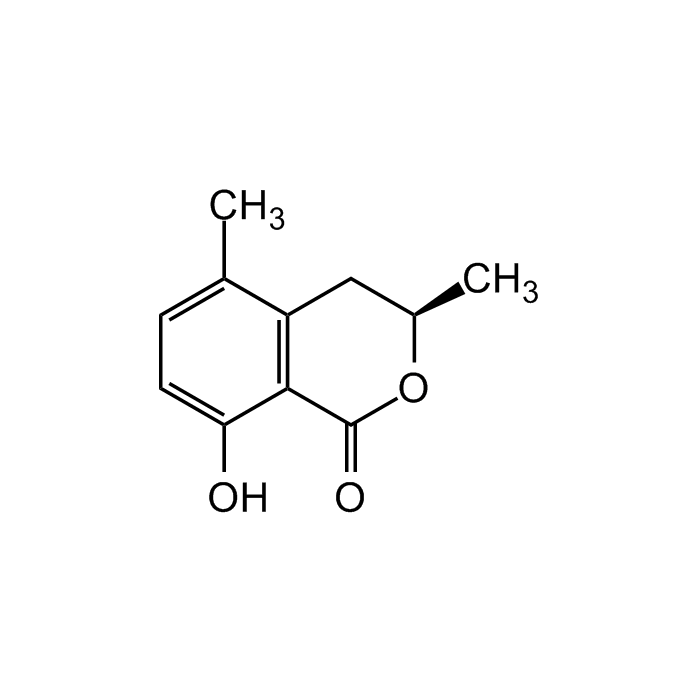
5-Methylmellein
As low as
85
CHF
CHF 85.00
In stock
Only %1 left
BVT-0413-M0011 mgCHF 85.00
| Product Details | |
|---|---|
| Synonyms | 5- Methylochracin; 3,4-Dihydro-8-hydroxy-3,5-dimethyl-1H-2-benzopyran1- one; 3,4-Dihydro-8-hydroxy-3,5-dimethylisocoumarin |
| Product Type | Chemical |
| Properties | |
| Formula |
C11H12O3 |
| MW | 192.2 |
| CAS | 7734-92-1 |
| Source/Host Chemicals | Isolated from Fusicoccum amygdalis. |
| Purity Chemicals | ≥98% (HPLC) |
| Appearance | Pale yellow powder. |
| Solubility | Soluble in DMSO, chloroform or dichloromethane. |
| Identity | Determined by 1H-NMR. |
| Declaration | Manufactured by BioViotica. |
| InChi Key | YETSBBYQOFXYGV-SSDOTTSWSA-N |
| Smiles | C[C@@H]1CC2=C(C(O)=CC=C2C)C(=O)O1 |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice | Protect from light when in solution. |
| Use/Stability |
Stable for at least 1 year after receipt when stored at -20°C. After reconstitution protect from light at -20°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
- Widespread fungal pentaketide.
- Weak antibacterial, antifungal and antiviral activity.
- Specific inhibitor of human DNA polymerase λ.
- Weak antigerminative activity.
Product References
- 5-Methylmellein, a new natural dihydroisocoumarin: A. Ballio, et al.; Tetrahedron Lett. 1966, 3723 (1966)
- (-)-5-Methylmellein and catechol derivatives from four semecarpus species: R.C. Carpenter, et al.; Phytochemistry 19, 445 (1980)
- 3-Methyl-3,4-dihydroisocoumarins and related compounds from the ascomycete family xylariaceae: J.R. Anderson & R.L. Edwards; J. Chem. Soc. Perkin Trans I 1983, 2185 (1983)
- Dihydroisocoumarins from fungi: isolation, structure elucidation, circular dichroism and biological activity: K. Krohn, et al.; Phytochemistry 45, 313 (1997)
- Nodulisporol and nodulisporone, novel specific inhibitors of human DNA polymerase λ from a fungus, Nodulisporium sp.: S. Kamisuki, et al.; Bioorg. Med. Chem. 15, 3109 (2007)
- Bioactive aromatic derivatives from endophytic fungus, Cytospora sp.: S. Lu, et al.; Nat. Prod. Commun. 6, 661 (2011)
- Guaiane sesquiterpenes from Biscogniauxia nummularia featuring potent antigerminative activity: S. Amand, et al.; J. Nat. Prod. 75, 798 (2012)