Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
Chemodex
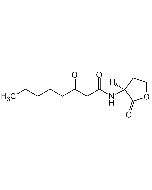
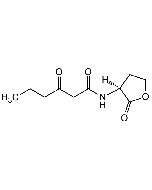
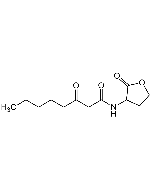
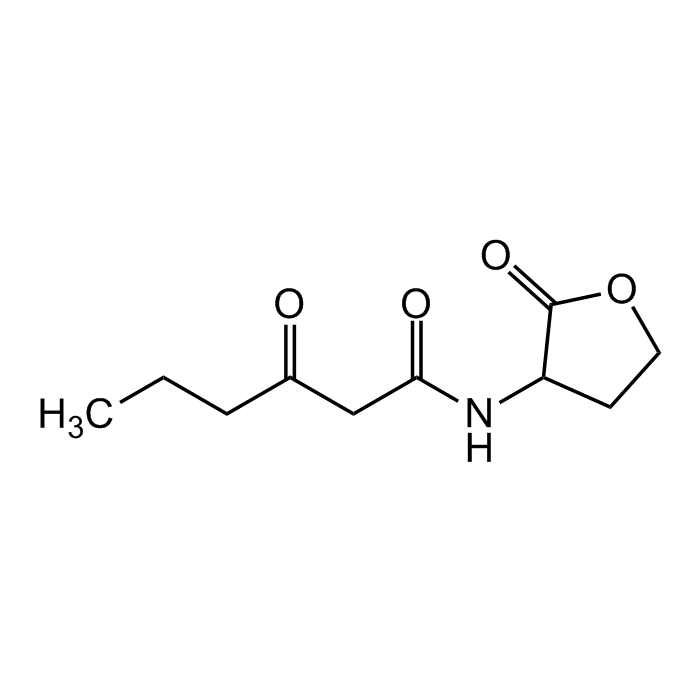
N-(3-Oxohexanoyl)-DL-homoserine lactone
| Product Details | |
|---|---|
| Synonyms | 3-oxo-C6-HSL; 3OC6-HSL; N-(β-Ketocaproyl)-DL-homoserine lactone |
| Product Type | Chemical |
| Properties | |
| Formula |
C10H15NO4 |
| MW | 213.2 |
| CAS | 76924-95-3 |
| Source/Host Chemicals | Synthetic. |
| Purity Chemicals | ≥97% (GC) |
| Appearance | White solid. |
| Solubility | Soluble in chloroform. Ethanol or methanol are not recommended as solvents as they have been shown to open the lactone ring. |
| Identity | Determined by NMR. |
| Declaration | Manufactured by Chemodex. |
| Other Product Data |
Click here for Original Manufacturer Product Datasheet |
| InChi Key | YRYOXRMDHALAFL-UHFFFAOYSA-N |
| Smiles | CCCC(=O)CC(=O)NC1CCOC1=O |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice |
Keep cool and dry. Protect from light and moisture. |
| Use/Stability | Stable for at least 2 years after receipt when stored at -20°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
N-(3-Oxohexanoyl)-DL-homoserine lactone is a small diffusible signaling molecule and is a member of N-acyl-homoserine lactone family. N-acylhomoserine lactones (AHL) are involved in quorum sensing, controlling gene expression, and cellular metabolism. The diverse applications of this kind of molecule include regulation of virulence in general, infection prevention, and formation of biofilms. At increased populations of bacteria, localized higher concentrations of 3OC6-HSL, an endogenous ligand to transcriptional factor LuxR, leads to increased production of both the AHL synthase and proteins responsible for bioluminescence. Numerous other species of bacteria also employ 3OC6-HSL in cell-to-cell communication. An autoinducer of P. fischeri luciferase and a specfic genetic regulator that is unrelated to at least one of the enzyme systems that it induces. It acts after excretion and accumulation in the extracellular medium.
(1) A. Eberhard, et al.; Arch. Microbiol. 146, 35 (1986) | (2) N.J. Bainton, et al.; Biochem. J. 288, 997 (1992) | (3) P. Williams, et al.; FEMS Microbiol. Lett. 100, 161 (1992) | (4) R.H. Gomer; Curr. Biol. 4, 734 (1994) | (5) A. Chatterjee, et al.; Appl. Environ.Microbiol. 61, 1959 (1995) | (6) Y.H. Dong, et al.; Nature 411, 813 (2001) | (7) R.L. Frey, et al.; Environ. Sci. Tech. 44, 7465 (2010)