Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
Chemodex
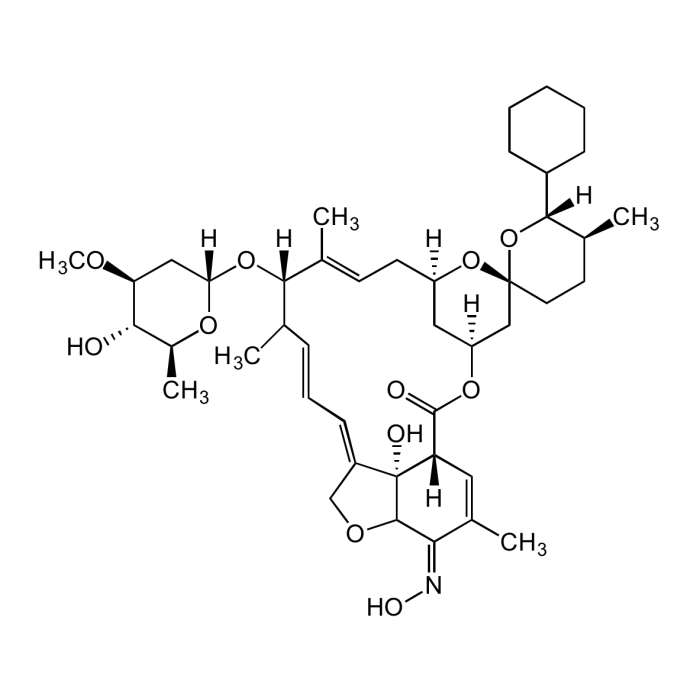
Selamectin
| Product Details | |
|---|---|
| Synonyms | Revolution; Stronghold; (5Z)-25-Cyclohexyl-4'-O-de(2,6-dideoxy-3-O-methyl-alp.-L-arabino-hexopyranosyl)-5-demethoxy-25-de(1-methylpropyl)-22,23-dihydro-5-(hydroxyimino)avermectin A1a |
| Product Type | Chemical |
| Properties | |
| Formula | C43H63NO11 |
| MW | 770.0 |
| CAS | 220119-17-5 |
| Source/Host Chemicals | Synthetic. |
| Purity Chemicals | ≥99% (HPLC) |
| Appearance | Off-white powder. |
| Solubility | Soluble in DMSO, dichloromethane, ethanol or methanol. Slightly soluble in water. |
| Identity | Determined by 1H-NMR. |
| Declaration | Manufactured by Chemodex. |
| Other Product Data |
Click here for Original Manufacturer Product Datasheet |
| InChi Key | AFJYYKSVHJGXSN-HNLAYDFISA-N |
| Smiles | O[C@@H]1[C@@H](OC)C[C@@](O[C@](C(C)/C=C/C=C(CO2)/[C@@](C2/C(C(C)=C3)=N\O)(O)[C@]3([H])C4=O)([H])/C(C)=C/C[C@]5([H])C[C@@](O4)([H])C[C@]6(CC[C@H](C)[C@](C7CCCCC7)([H])O6)O5)([H])O[C@H]1C |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +20°C |
| Long Term Storage | +4°C |
| Handling Advice |
Keep cool and dry. Protect from light and moisture. |
| Use/Stability | Stable for at least 2 years after receipt when stored at +4°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Avermectin derivative prepared by hydrolysis and oximation of doramectin. Selamectin is a potent nematocide used for the treatment of parasites that acts as agonist of the GABA (gamma-aminobutyric acid) neurotransmitter in nerve cells and also binds to glutamate-gated chloride channels in nerve and muscle cells of invertebrates. In both cases it blocks the transmission of neuronal signals of the parasites, which are either paralyzed and expelled out of the body, or they starve. It also affects the reproduction of some parasites by diminishing oviposition or inducing an abnormal oogenesis. The substance fights both internal and surface parasitic infection. Compound can be used as analytical reference material.
Antiviral effective against 2019-nCoV
(1) B.F. Bishop, et al.; Vet. Parasitol. 91, 163 (2000) | (2) R.H. Six, et al.; Vet. Parasitol. 91, 321 (2000) | (3) B.J. Banks, et al.; Bioorg. Med. Chem. 8, 2017 (2000) | (4) J. Griffin, et al.; J. Vet. Pharmacol. Ther. 28, 257 (2005) | (5) M.U. Esatgil, et al.; Pol. J. Vet. Sci. 11, 67 (2008) | (6) M.U. Esatgil, et al.; Pol. J. Vet. Sci. 11, 143 (2008) | (7) J. Geyer, et al.; J. Vet. Pharmacol. Ther. 32, 87 (2009) | (8) A. Melotti, et al.; EMBO Mol. Med. 6, 1263 (2014)
Repurposing of clinically approved drugs for treatment of coronavirus disease 2019 in a 2019-novel coronavirus-related coronavirus model: H.H. Fan, et al.; Chin. Med. J. 133, 1051 (2020)





