Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
Chemodex
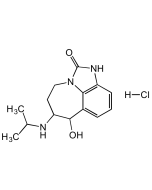
(±)-Verapamil hydrochloride

| Product Details | |
|---|---|
| Synonyms | Manidon |
| Product Type | Chemical |
| Properties | |
| Formula | C27H38N2O4 . HCl |
| MW | 491.06 |
| CAS | 152-11-4 |
| RTECS | YV8320000 |
| Source/Host Chemicals | Synthetic |
| Purity Chemicals | ≥99% (Titration) |
| Appearance | White powder. |
| Solubility | Soluble in methanol (50 mg/ml), ethanol or water. |
| Identity | Determined by NMR. |
| Declaration | Manufactured by Chemodex. |
| Other Product Data |
Click here for Original Manufacturer Product Datasheet |
| InChi Key | DOQPXTMNIUCOSY-UHFFFAOYSA-N |
| Smiles | Cl.COC1=C(OC)C=C(CCN(C)CCCC(C#N)(C(C)C)C2=CC(OC)=C(OC)C=C2)C=C1 |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | +4°C |
| Handling Advice |
Keep cool and dry. Protect from light and moisture. |
| Use/Stability | Stable for at least 2 years after receipt when stored at +4°C. |
| Documents | |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Verapamil hydrochloride is an α-adrenergic receptor (α-AR) antagonist and calcium channel protein inhibitor that blocks the L-type Ca2+ channels in smooth and cardiac muscle cells. Verapamil is an antiarrhythmic agent and vasodilator known to reduce the renal clearance of digoxin and induce apoptosis in primary and metastatic colon adenocarcinoma human cell lines in vitro. It has been observed that verapamil can induce currents by itself, presumably by acting on the potassium and chloride leakage. Verapamil has also been used as an inhibitor of drug efflux pump proteins such as Mdr (P-glycoprotein). Verapamil is a substrate of CYP3A4 and CYP2C6. It is also used in fluorescent cell sorting for DNA content, as it blocks efflux of a variety of DNA-binding fluorophores such as Hoechst 33342. Recent research has shown verapamil to be an effective treatment for diabetes in animal models. Verapamil helps treat diabetes by limiting TXNIP expression.
(1) A. Fleckenstein; Annu. Rev. Pharmacol. Toxicol. 17, 149 (1977) | (2) I.B. Shchepotin, et al.; Anticancer Res. 14, 1027 (1994) | (3) B.B. Lonsberry, et al.; Pharmacol. 49, 23 (1994) | (4) W.T. Bellamy; Annu. Rev. Pharmacol. Toxicol. 36, 161 (1996) | (5) T. Kantola, et al.; Clin. Pharmacol. Ther. 64, 177 (1998) | (6) M. Verschraagen, et al.; Pharmacol. Res. 40, 301 (1999) | (7) G. Xu, et al.; Diabetes. 61, 848 (2012)