Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
Chimerigen
IL-23 (mouse):Fc (human) (rec.)

| Product Details | |
|---|---|
| Synonyms | Interleukin-23 |
| Product Type | Protein |
| Properties | |
| Source/Host | CHO cells |
| Sequence |
The mouse IL-23 complex composed of the p40 subunit (aa 23-335) and the p19 subunit (aa 20-196) is fused through a polypeptide linker to the Fc region of human IgG1. |
| Crossreactivity | Mouse |
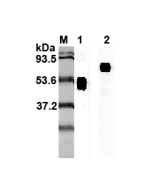
| Purity | ≥98% (SDS-PAGE) |
| Endotoxin Content | <0.06EU/μg protein (LAL test; Lonza). |
| Reconstitution | Reconstitute at 100μg/ml in sterile PBS. |
| Formulation | Lyophilized from 0.2μm-filtered solution in PBS. |
| Protein Negative Control | |
| Other Product Data |
NCBI reference P43432: p40 subunit (mouse) |
| Declaration | Manufactured by Chimerigen. |
| Shipping and Handling | |
| Shipping | BLUE ICE |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice |
Avoid freeze/thaw cycles. Centrifuge lyophilized vial before opening and reconstitution. |
| Use/Stability |
Stable for at least 1 year after receipt when stored at -20°C. Working aliquots are stable for up to 3 months when stored at -20°C. |
| Documents | |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Interleukin-23 (IL-23) is a heterodimeric cytokine composed of two disulfide-linked subunits, a p19 subunit that is unique to IL-23, and a p40 subunit that is shared with IL-12 (1-5). Although p19 is expressed by activated macrophages, dendritic cells, T cells, and endothelial cells, only activated macrophages and dendritic cells express p40 concurrently to produce IL-23. The functional IL-23 receptor complex consists of two receptor subunits, the IL-12 receptor β 1 subunit (IL-12 Rβ1) and the IL-23-specific receptor subunit (IL-23 R). IL-23 has biological activities that are similar to, but distinct from IL-12. Both IL-12 and IL-23 induce proliferation and IFN-γ production by human T cells. While IL-12 acts on both naie and memory human T cells, the effects of IL-23 is restricted to memory T cells. In mouse, IL-23 but not IL-12, has also been shown to induce memory T cells to secret IL-17, a potent proinflammatory cytokine. IL-12 and IL-23 can induce IL-12 production from mouse splenic DC of both the CD8-and CD8+ subtypes, however only IL-23 can act directly on CD8+ DC to mediate immunogenic presentation of poorly immunogenic tumor/self peptide.
- Myeloid Cell–Intrinsic IRF5 Promotes T Cell Responses through Multiple Distinct Checkpoints In Vivo, and IRF5 Immune-Mediated Disease Risk Variants Modulate These Myeloid Cell Functions: J. Yan, et al.; J. Immunol. 205, 1024 (2020)