Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
Innaxon
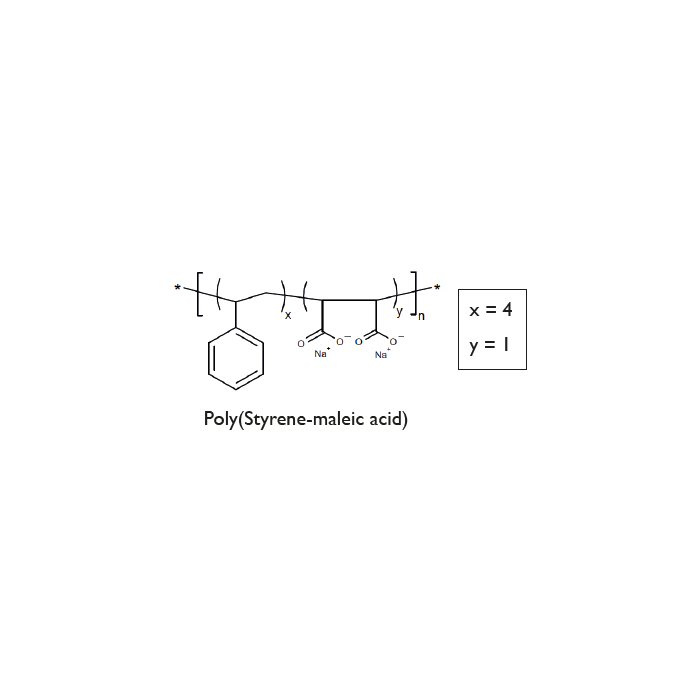
SMA-400
| Product Details | |
|---|---|
| Synonyms | Lipodisq™ Styrene:Maleic Acid Copolymer 4:1 |
| Product Type | Chemical |
| Properties | |
| Formula |
C36H34O4Na2 |
| MW |
10,500 (based on weight) 4,500 (based on number) |
| CAS | 26762-29-8 |
| Purity Chemicals | >98% |
| Appearance | White powder. |
| Solubility | Soluble in water, and buffer solutions (pH 8.50-10.50) to allow the formulation of a proprietary, thermostable, aqueous lipid nanoparticle. |
| Reconstitution | Avoid the use of buffers with divalent ions such as Ca or Mg or pH <8.5 or >10.5, which can cause P(SMA) precipitation or interfere with SMA-Lipid Particle fornulation or stability. |
| Formulation | Lipodisq™ are nanosized lipid-based discoidal particles that can be manufactured with P(SMA)/SMALP and lipids such as DMPC (14:0 PC) (DMPC: IAX-700-400) to incorporate hydrophobic, poorly water-soluble active compounds. |
| Declaration | Manufactured by Innaxon. |
| Other Product Data |
Click here for Original Manufacturer Product Datasheet: Our product description may differ slightly from the original manufacturer's product datasheet. Lipodisq™ technology is covered by one or more of the following patents owned by Malvern Cosmeceutics Limited: AU2006253886, CA2611144, CN101184473B, EP1890675, GB2426703, IN261468, JP5142898, US8623414 and WO/2021/005340A1 pending. The purchaser is licensed under those patents to use these assemblies for the purpose of research and development only, but not for the purpose of delivery of agents for clinical use to humans or veterinary use to animals for therapeutic, diagnostic or prophylactic purposes, which uses are specifically prohibited. |
| Smiles | |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +20°C |
| Long Term Storage | +20°C |
| Handling Advice |
Do not freeze. Keep sterile. Avoid skin and eye contact. |
| Use/Stability | Stable for at least 2 years after receipt when stored at +20°C. |
| Documents | |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
- A nanoparticle (11-40nm) drug delivery system comprising a discoidal phospholipid bilayer membrane stabilized by a chaperone molecular annulus.
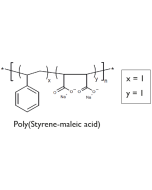
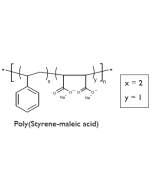
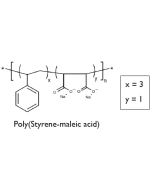
- Lipodisq™ polymers are available as 4 defined structures (1:1, 2:1, 3:1, and 4:1 Styrene to MaleicAcid (SMA) rations) each individually operational within a selected pH range for optimal working conditions.
- Components are batch-tested for Lipodisq™ formation using buffer systems available, which are tested for nano-formulated drug analysis by Dynamic Light Scattering (DLS). These buffer solutions are endotoxin-tested and sterile.
- Lipodisq™ formation is highly efficient and nanodiscs show a good safety profile and are suitable for in vitro and in vivo (in experimental animals) investigations.
- Lipodisq™ are nanosized lipid-based discoidal particles to incorporate hydrophobic, poorly water-soluble active compounds, such as peptides, lipids, lipoproteins, transmembrane proteins and glycolipids. Applications of Lipodisq™ include functional and structural characterization of the cargo and drug delivery with improved bioavailability, and biological half-life in vivo (PD/PK) or delivery of antigens preserved in their native conformation for immunization purposes.
Answering the call for Membrane Protein (MP) and MP reconstituting membrane simulants which do not necessitate the use of mediating detergent, is a copolymer of styrene and maleic anhydride subsequently hydrolyzed into amphipathic polystyrene-co-maleic acid (SMA). Nanodiscs resulting from SMA are also referred to as styrene-maleic acid-lipid particles (SMALP). SMA-lipid particles are formed by directly extracting membrane proteins either from native cellular membranes (giving native nanodiscs) or from an intermediary MP-reconstituted synthetic membrane system to ultimately form self-assembled nanodisc structures of a general 10-12 nm diameter. The demonstration of SMA-lipid particles as a monodisperse MP reconstitution system was reported in 2009, although the interaction of SMA with phospholipids to generate disc-shaped structures (now known as empty lipid nanodiscs), was previously established and investigated for use as a drug delivery system years preceding this discovery.
At a physiological pH of 7-8, SMA monomer ratios of 2:1 and 3:1 are most commonly used due to their optimal efficiency for nanodisc extraction from phospholipid bilayers. The ability of SMA to create monodisperse nanodiscs with size flexibility facilitates the reconstitution of a range of oligomeric MPs and MP complexes for analysis by various studies including fluorescence microscopy, NMR, and single-particle cryo-EM. Whereas in a statistical version of SMA, like the commercial Malvern polymer Lipodisq™, monomers are evenly distributed throughout the polymer chain sequence in proportion to their ratio as well as exhibiting greater dispersity in chain length.
Published procedures using a ratio of 2:1 (w/w) for P(SMA)/SMA polymer and phospholipid, such as 100mg P(SMA)/SMA polymer and 50mg DMPC. Selected hydrophobic drug candidates or peptides or transmembrane proteins (MP) are mixed with the aqueous phospholipid emulsion
making up 2.5%. This is stirred at temperatures above the phase transition temperature of the lipid (>24°C for DMPC) before aqueous P(SMA)/SMA polymer at 5% is added drop-wise and with pauses until an approximate volume ratio of 1:1 is reached and the lipid emulsion clears.
- Alternatively, P(SMA)/SMA polymer solutions are mixed with native (cell or bacterial) membranes to form native nanodiscs.
- Stirring time, pH, selected buffer type and strength (e.g. HEPES, NaCl, TRIS or PBS w/o Ca++ and Mg++) and temperature of the phospholipid emulsion containing the MP or active ingredient, need to be optimized.
- Further purification of formed LipodisqTM can be achieved by ultracentrifugation at >100,000 x g to remove residual lipid, surplus P(SMA)polymer with the Lipodisq™ nanodiscs remaining in the supernatant. Alternatively, size exclusion (SEC) methods can also be applied.
- Reconstitution of membrane proteins: a GPCR as an example: A.D. Goddard, et al.; Methods Enzymol. 556, 405 (2015)
- The styrene-maleic acid copolymer: a versatile tool in membrane research: J.M. Doerr, et al.; Eur. Biophys. J. 45, 3 (2016)
- From polymer chemistry to structural biology: The development of SMA and related amphipathic polymers for membrane protein extraction and solubilization: J.F. Bada Juarez, et al.; Chem. Phys. Lipids. 221, 167 (2019)
- Effects of charged lipids on the physicochemical and biological properties of lipid-styrene-maleic acid copolymer discoidal particles: M. Tanakaa, et al.; Biochim. Biophys. Acta. Biomembr. 1862, 183209 (2020)
- Physicochemical Characterization, Toxicity and In Vivo Biodistribution Studies of a Discoidal, Lipid-Based Drug Delivery Vehicle: Lipodisq Nanoparticles Containing Doxorubicin: M.L. Torgersen, et al.; J. Biomed. Nanotechnol. 16, 41 (2020)
- Understanding the Structural Pathways for Lipid Nanodisc Formation: How Styrene Maleic Acid Copolymers Induce Membrane Fracture and Disc Formation: V.A. Bjornestad, et al.; Langmuir 37, 6178 (2021)
- Applications of Synthetic Polymer Discoidal Lipid Nanoparticles to Biomedical Research: M. Tanaka; Chem. Pharm. Bull. 70, 507 (2022)
- Mechanisms of Formation, Structure, and Dynamics of Lipoprotein Discs Stabilized by Amphiphilic Copolymers: A Comprehensive Review: P.S. Orekhov, et al.; Nanomaterials 12, 361 (2022)