Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
SouthBayBio
SUMO2 (human) (rec.) (Rhodamine 110)
| Product Details | |
|---|---|
| Synonyms | Small Ubiquitin-related Modifier 2; HSMT3; SMT3 Homolog 2; Sentrin-2; Ubiquitin-like Protein SMT3A |
| Product Type | Protein |
| Properties | |
| Source/Host | E. coli |
| Sequence | Human SUMO2 (aa1-93) (Accession Nr. P61956) conjugated at the C-terminus to a quenched Rhodamine 110 dye. |
| Crossreactivity | Human |
| Application | Protein-based substrate. Typical working concentration range is 50-500nM. Hydrolysis of the conjugate results in fluorescence observable by excitation at 485nm and emission at 535nM. |
| Label/Conjugates | Rhodamine |
| MW | ~11kDa |
| Purity | ≥97% (LCMS) |
| Concentration | Lot dependent. |
| Accession Number | P61956 |
| Formulation | Liquid. In 50mM HEPES pH 7.5, 100mM sodium chloride. |
| Other Product Data |
Click here for a Typical Lot-specific Product Datasheet from the Original Manufacturer |
| Declaration | Manufactured by South Bay Bio. |
| Shipping and Handling | |
| Shipping | DRY ICE |
| Short Term Storage | -80°C |
| Long Term Storage | -80°C |
| Handling Advice |
Aliquot to avoid freeze/thaw cycles. Protect from light. |
| Use/Stability | Stable for at least 1 year after receipt when stored at -80°C. |
| Documents | |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
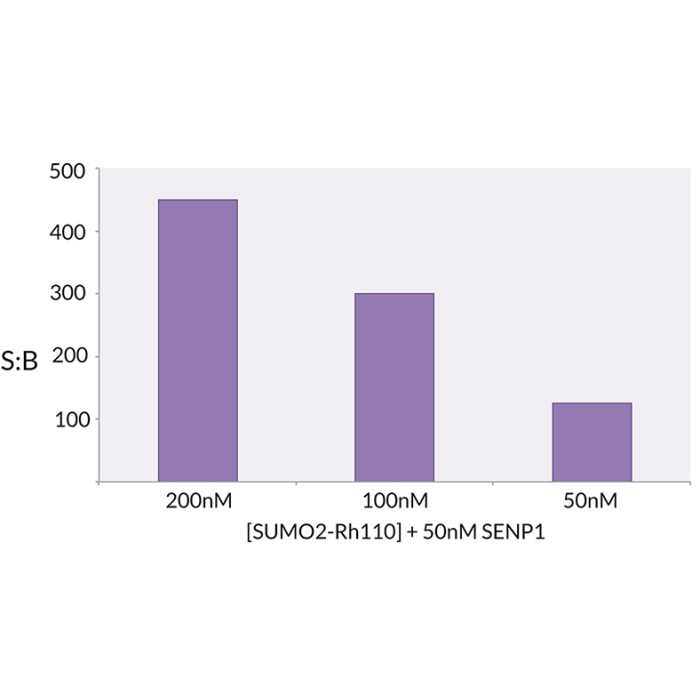
SUMO2 is a Ubiquitin-like protein (UBL) that can be covalently attached to proteins as a monomer or as a lysine-linked polymer. Covalent attachment via an isopeptide bond to its substrates requires prior activation by the E1 complex SAE1-SAE2 and linkage to the E2 enzyme UBE2I, and can be promoted by an E3 ligase such as PIAS1-4, RANBP2, CBX4 or ZNF451. This post-translational modification on lysine residues of proteins plays a crucial role in a number of cellular processes such as nuclear transport, DNA replication and repair, mitosis and signal transduction. This SUMO2 substrate is C-terminally derivatized with a bis-Gly-Rhodamine 110 fluorophore. The bis-Gly-Rh110 is quenched until the amide bond between the C-terminal glycine and the rhodamine compound is hydrolyzed. The efficiency of quenching combined with the powerful signal upon hydrolysis yields an unparalleled signal-to-background. SUMO2-Rh110 can be used to study the deSUMOylating activity of hydrolases SENP1 and SENP2, among other deSUMOylating enzymes. The substrate activity of SUMO2-Rhodamine 110 was determined by measuring the SENP1 catalyzed release of unquenched Gly-Rh-110.