Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
SynKinase
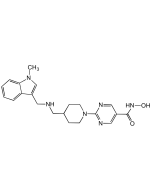
Vorinostat
As low as
57
CHF
CHF 57.00
In stock
Only %1 left
SYN-3006-M0055 mgCHF 57.00
SYN-3006-M01010 mgCHF 71.00
SYN-3006-M05050 mgCHF 99.00
SYN-3006-M100100 mgCHF 128.00

| Product Details | |
|---|---|
| Synonyms | SAHA; Suberoyl anilide bishydroxamide; Suberoylanilide hydroxamic acid; |
| Product Type | Chemical |
| Properties | |
| Formula | C14H20N2O3 |
| MW | 264.3 |
| CAS | 149647-78-9 |
| Purity Chemicals | ≥95% |
| Appearance | Solid. |
| Solubility | Soluble in DMSO or ethanol. Almost insoluble in water. |
| Identity | Determined by 1H-NMR. |
| Declaration | Manufactured by SynKinase. |
| Other Product Data |
Click here for Original Manufacturer Product Datasheet Our product description may differ slightly from the original manufacturers product datasheet. |
| InChi Key | WAEXFXRVDQXREF-UHFFFAOYSA-N |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Use/Stability | Stable for at least 2 years after receipt when stored at -20°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
Vorinostat (SAHA, MK0683, Zolinza) inhibits the enzymatic activity of histone deacetylases HDAC1, HDAC2 and HDAC3 (Class I) and HDAC6 (Class II) at nanomolar concentrations (IC50< 86 nM). These enzymes catalyze the removal of acetyl groups from the lysine residues of histones proteins. In some cancer cells, there is an overexpression of HDACs, or an aberrant recruitment of HDACs to oncogenic transcription factors causing hypoacetylation of core nucleosomal histones. By inhibiting histone deacetylase, vorinostat causes the accumulation of acetylated histones and induces cell cycle arrest and/or apoptosis of some transformed cells. The mechanism of the antineoplastic effect of vorinostat has not been fully characterized.
Product References
- Vorinostat, a histone deacetylase inhibitor, enhances the response of human tumor cells to ionizing radiation through prolongation of gamma-H2AX foci: A. Munshi, et al.; Mol. Cancer Ther. 5, 1967 (2006)
- Poly(beta-amino ester) nanoparticles enable tumor-specific TRAIL secretion and a bystander effect to treat liver cancer: H.J. Vaughan, et al.; Mol. Ther. Oncolytics 21, 377 (2021)