Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
AdipoGen Life Sciences
Gasdermin E (human) ELISA Kit
| Product Details | |
|---|---|
| Synonyms | GSDME; DFNA5; ICERE-1; Inversely Correlated with Estrogen Receptor Expression 1 |
| Product Type | Kit |
| Properties | |
| Application Set | Quantitative ELISA |
| Specificity |
Detects human Gasdermin E (C-terminal and full-length) in serum, plasma and cell culture supernatant. |
| Crossreactivity | Human |
| Quantity |
1 x 96 wells |
| Sensitivity | 50pg/ml |
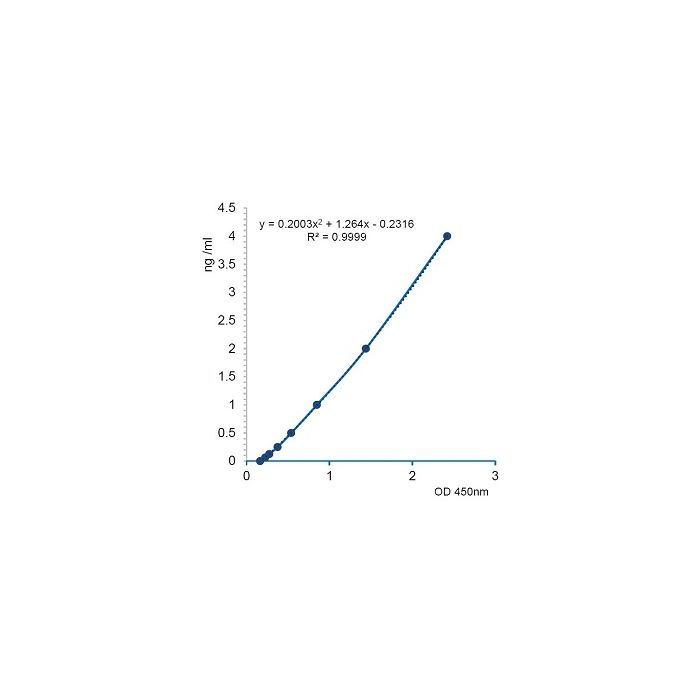
| Range | 0.0625 to 4ng/ml |
| Sample Type |
Cell Culture Supernatant Plasma Serum |
| Assay Type | Sandwich |
| Detection Type | Colorimetric |
| Other Product Data |
UniProt link O60443: Gasdermin E (human) |
| Accession Number | O60443 |
| Shipping and Handling | |
| Shipping | BLUE ICE |
| Short Term Storage | +4°C |
| Long Term Storage | +4°C |
| Handling Advice |
After standard reconstitution, prepare aliquots and store at -20°C. Avoid freeze/thaw cycles. Plate and reagents should reach room temperature before use. |
| Use/Stability | 18 months after the day of manufacturing. See expiry date on ELISA Kit box. |
| Documents | |
| Manual |
 Download PDF Download PDF |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Programmed cell death is a key mechanism involved in several biological processes ranging from development and homeostasis to immunity. Apoptosis, pyroptosis and NETosis are three well-studied modes of cell death. Pyroptosis is a form of necrotic cell death that has emerged as an important innate immune mechanism against intracellular pathogens. All three modes of cell death cross-talk between them and converge onto the activation of the same cell death effector, the pore-forming protein Gasdermin D (GSDMD). The inflammasome platform leads to activation of caspase-1 and also the recently characterized caspase-11 (non-canonical inflammasome pathway), leading to cleavage of the intracellular protein Gasdermin D. Upon caspase-1/11 cleavage of the Gasdermin N- and C-domain linker, the cleaved N-terminal fragment of Gasdermin D oligomerizes and forms pores on the host cell membrane, leading to a cell death called pyroptosis. The Gasdermin family is an important protein that mediates pyroptosis, it can induce cell death and promote the release of inflammatory factors. It is mainly composed of six members: Gasdermin A (GSDMA), Gasdermin B (GSDMB), Gasdermin C (GSDMC), GSDMD, Gasdermin E (GSDME; also called deafness, autosomal dominant 5, DFNA5), and DFNB58. Recent studies found that GSDME features a caspase-3 /-7 cleavage motif in its linker region. Similar to GSDMD, cleavage of GSDME by caspases-3 /-7 liberates the N-terminal pyroptosis-inducing domain (GSDMENT) from its autoinhibitory C-terminal regulatory domain to trigger membrane pores and pyroptosis. Gasdermin E is expressed in different tissues /cells such as placenta, brain, heart, kidney, cochlea, intestines, and IgE-primed mast cells. Specific cells express GSDME, and in such cells caspase-3 activation is switched from driving an apoptotic program to causing pyroptosis. Thus, certain cells are programmed to undergo pyroptosis instead of apoptosis. GSDME-knockout mice were shown to be resistant to the toxicity of chemotherapy drugs such as cisplatin. GSDME-N terminal domain also permeabilizes the mitochondrial membrane, releasing cytochrome C and activating the apoptosome. Recently it has been reported that granzyme B from killer cells activates caspase-independent pyroptosis in target cells by directly cleaving GSDME at the same site as caspase-3. Mutations in GSDME in human are associated with development of heritable, nonsyndromal deafness. GSDME expression is suppressed in many cancers and reduced GSDME is associated with decreased breast cancer survival. GSDME gene expression has been shown to be associated with favorable prognosis following chemotherapy, and it could therefore be a potential predictive biomarker.
The Gasdermin E (human) ELISA Kit (AG-45B-0024) detects the C-terminal domain of human Gasdermin E as well as the full-length protein. Upon cleavage of Gasdermin E and pore formation, the C-terminus fragment should be released in the extracellular space and found in serum / plasma. The role of the secreted C-terminus fragment of Gasdermin protein family has not been characterized yet.