Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
AdipoGen Life Sciences
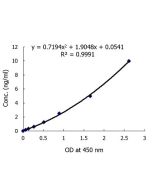
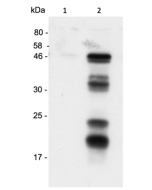
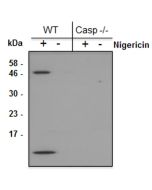
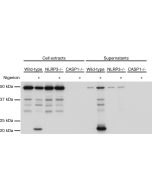
Gasdermin D (mouse) ELISA Kit
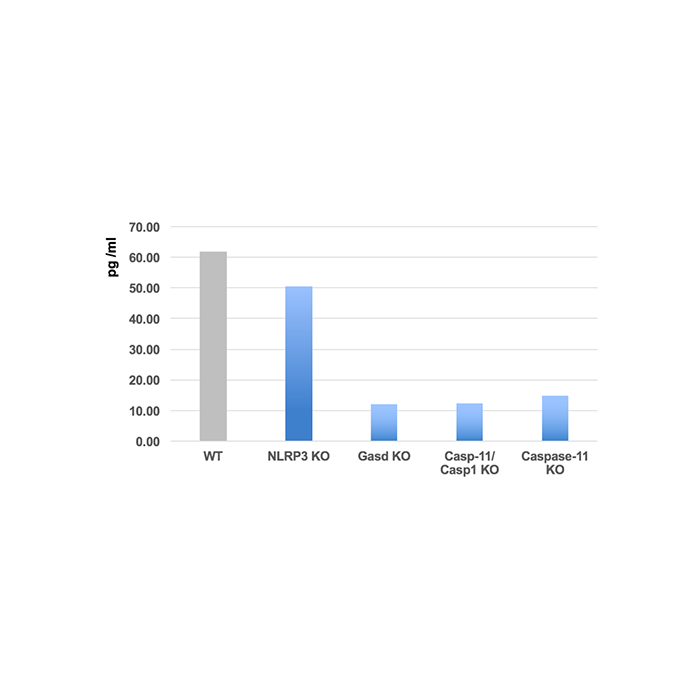
Method: Bone marrow-derived macrophages (BMDMs) are transfected for 4h with LPS in different KO mice strains. Supernatants are analysed.
| Product Details | |
|---|---|
| Synonyms | Gasdermin Domain-containing Protein 1; Gasdermin-D (C-terminal); GSDMD-CT; Gsdmd |
| Product Type | Kit |
| Properties | |
| Application Set | Quantitative ELISA |
| Specificity |
Detects full-length and cleaved mouse Gasdermin D (C-terminus) in cell extracts and mainly C-terminus Gasdermin D in cell culture supernatants. Does not cross-react with human or rat gasdermin D. |
| Crossreactivity | Mouse |
| Quantity |
1 x 96 wells |
| Sensitivity | 14pg/ml |
| Range | 15.6 to 1000pg/ml |
| Sample Type |
Cell Culture Supernatant Cell Lysate Serum |
| Assay Type | Sandwich |
| Detection Type | Colorimetric |
| Other Product Data |
UniProt link Q9D8T2: Gasdermin D (mouse) Also available: EMBO Inflammasome Symposia 2018 - Official Gasdermin D (mouse) ELISA Kit Poster! for download |
| Accession Number | Q9D8T2 |
| Shipping and Handling | |
| Shipping | BLUE ICE |
| Short Term Storage | +4°C |
| Long Term Storage | +4°C |
| Handling Advice |
After standard reconstitution, prepare aliquots and store at -20°C. Avoid freeze/thaw cycles. Plate and reagents should reach room temperature before use. |
| Use/Stability | 12 months after the day of manufacturing. See expiry date on ELISA Kit box. |
| Documents | |
| Manual |
 Download PDF Download PDF |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Inflammasomes are multimeric protein complexes that comprise a sensor (e.g. NLRP3), an adaptor (ASC/Pycard) and the procaspase-1. An inflammasome assembles in response to a diverse range of pathogen-associated or danger-associated molecular patterns (PAMPs or DAMPs). The inflammasome platform leads to activation of caspase-1, which further induces maturation of interleukin-1β and -18 (IL-1β and IL-18) through proteolytic cleavage of pro-IL-1β and pro-IL-18. Activated caspase-1, and also the recently characterized caspase-11 non-canonical inflammasome pathway, also cleave the intracellular gasdermin D, which leads to a particular form of inflammatory cell death called pyroptosis. The gasdermin family members contain N-terminal domains that are capable of forming membrane pores to induce cytolysis, whereas the C-terminal domains of gasdermins function as inhibitors of such cytolysis through intramolecular domain association. Caspase-1 or -11 cleavage of gasdermin D is required for regulation of pyroptosis: upon protease cleavage of the gasdermin N- and C-domain linker, the disruption of the intramolecular domain interaction in the presence of lipids releases the N-domain to assemble oligomeric membrane pores that trigger cell death. Gasdermin D seems to be a key effector in the LPS-induced lethal sepsis.
- Dynamin-related protein 1 deficiency accelerates lipopolysaccharide-induced acute liver injury and inflammation in mice: L. Wang, et al.; Nature Commun. Biol. 4, 894 (2021) [Samples: Serum, Cell Lysates]
- Damage-induced pyroptosis drives endogenous thymic regeneration via induction of Foxn1 by purinergic receptor activation: S. Kinsella, et al.; bioRxiv doi 10.1101/2023.01.19.524800v1 (2023)