Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
AdipoGen Life Sciences
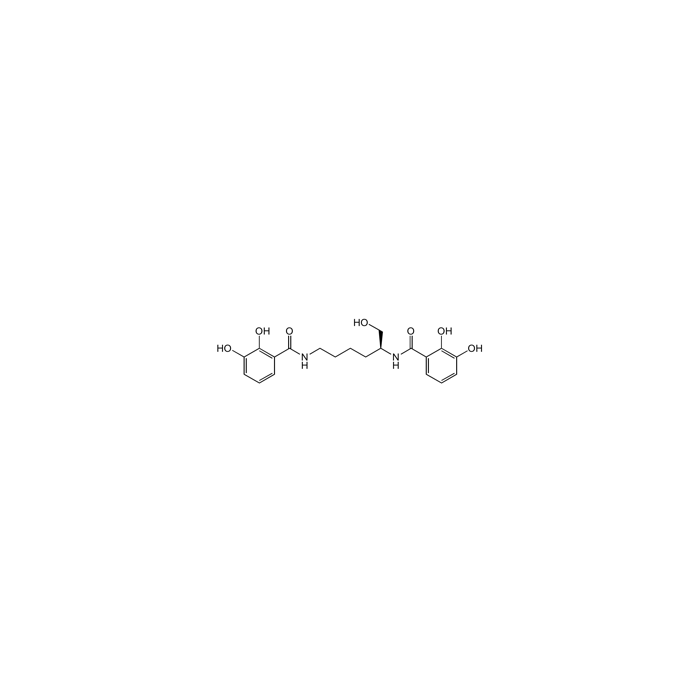
Myxochelin A
As low as
120
CHF
CHF 120.00
In stock
Only %1 left
AG-CN2-0470-C100100 µgCHF 120.00
AG-CN2-0470-M0011 mgCHF 420.00
| Product Details | |
|---|---|
| Synonyms | (S)-N,N'-(6-Hydroxyhexane-1,5-diyl)bis(2,3-dihydroxybenzamide) |
| Product Type | Chemical |
| Properties | |
| Formula |
C20H24N2O7 |
| MW | 404.4 |
| CAS | 120243-02-9 |
| Source/Host Chemicals | Synthetic. Originally isolated from Pyxidicoccus fallax HKI 727. |
| Purity Chemicals | ≥99% (NMR) |
| Appearance | Solid. |
| Solubility | Soluble in DMSO, ethanol, methanol, acetone, ethylacetate or chloroform. Insoluble in water or hexane. |
| Identity | Determined by 1H-NMR. |
| InChi Key | WNBVGSNESTZACV-LBPRGKRZSA-N |
| Smiles | OC[C@H](CCCCNC(=O)C1=C(O)C(O)=CC=C1)NC(=O)C1=CC=CC(O)=C1O |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice | Protect from light and moisture. |
| Use/Stability |
Stable for at least 2 years after receipt when stored at -20°C. Stable for at least 1 year after receipt when stored at +4°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
- Potent inhibitor of human 5-lipoxygenase (5-LO). This enzyme catalyzes two initial steps in the conversion of arachidonic acid into leukotrienes, well known mediators of inflammatory and allergic reactions.
- Iron-chelating compound.
- Anticancer antibiotic. Shown to inhibit tumor cell invasion in vitro.
- Antibacterial compound.
- Antioxidant with free radical scavenging activities.
Product References
- Myxochelin A, a new iron-chelating compound from Angiococcus disciformis (Myxobacterales). Production, isolation, physico-chemical and biological properties: B. Kunze, et al.; J. Antibiot. 42, 14 (1989)
- Myxochelin A, a cytotoxic antibiotic from the myxobacterium Angiococcus disciformis: J.W. Ahn, et al.; OPEM 2, 64 (2001)
- Antioxidative and free radical scavenging activities of Myxochelin A isolated from Angiococcus sp.(Myxobacteria): H.-H. Lee, et al.; Food Sci. Biotech. 11, 184 (2002)
- Absolute configuration and antitumor activity of myxochelin A produced by Nonomuraea pusilla TP-A0861: S. Miyanaga, et al.; J. Antibiot. 59, 698 (2006)
- Synthesis and evaluation of myxochelin analogues as antimetastatic agents: S. Miyanaga, et al.; Bioorg. Med. Chem. 17, 2724 (2009)
- Myxochelins target human 5-lipoxygenase: S. Schieferdecker, et al.; J. Nat. Prod. 78, 335 (2015)
- Harnessing enzymatic promiscuity in Myxochelin biosynthesis for the production of 5-Lipoxygenase inhibitors: J. Korp, et al.; Chembiochem. 16, 2445 (2015)