Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
AdipoGen Life Sciences
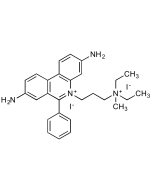
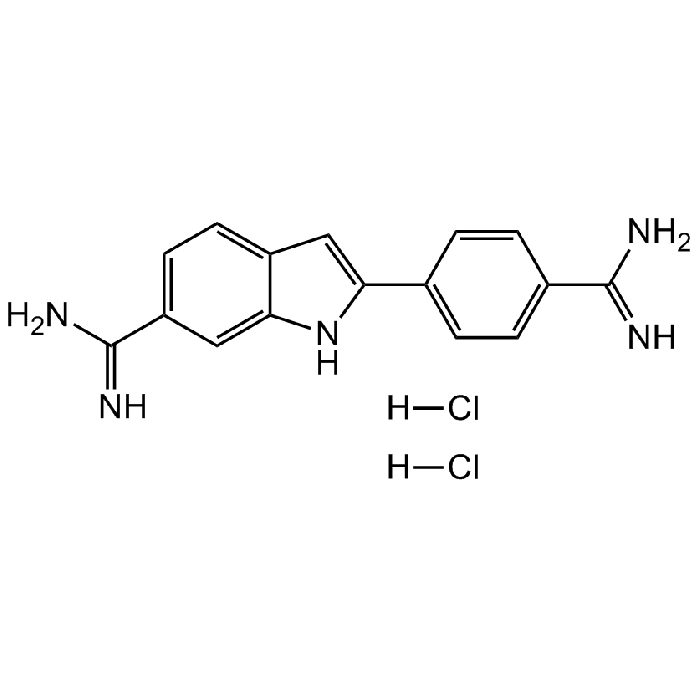
DAPI . dihydrochloride
As low as
45
CHF
CHF 45.00
In stock
Only %1 left
AG-CR1-3668-M0055 mgCHF 45.00
AG-CR1-3668-M02525 mgCHF 155.00
AG-CR1-3668-M100100 mgCHF 450.00
| Product Details | |
|---|---|
| Synonyms | 4',6-Diamidino-2-phenylindole dihydrochloride; 2-(4-Amidinophenyl)-6-indolecarbamidine dihydrochloride; FxCycle Violet |
| Product Type | Chemical |
| Properties | |
| Formula |
C16H15N5 . 2HCl |
| MW | 277.3 . 72.9 |
| CAS | 28718-90-3 |
| RTECS | NL5995050 |
| Purity Chemicals | ≥97% (HPLC) |
| Appearance | Yellow solid. |
| Solubility | Soluble in water (10mg/ml). May require heating and sonication. Slightly soluble in DMSO, ethanol, methanol or PBS. |
| Identity | Determined by 1H-NMR. |
| Other Product Data |
Note: Stock solutions (1mg/ml) should be stored in the dark and are stable in the dark at 2-8 °C for 1-2 weeks. For long-term storage, the solutions can be divided into aliquots and stored at -20 °C. |
| InChi Key | FPNZBYLXNYPRLR-UHFFFAOYSA-N |
| Smiles | [H]Cl.[H]Cl.NC(C1=CC=C2C(NC(C3=CC=C(C(N)=N)C=C3)=C2)=C1)=N |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice |
Keep cool and dry. Protect from light and moisture. |
| Use/Stability |
Stable for at least 2 years after receipt when stored at -20°C. Store solutions at -20°C in the dark. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
- Cell permeable fluorescent DNA stain. AT-sequence-specific DNA intercalator.
- Binds to the minor groove of double-stranded DNA (preferentially to adenine and thymine (AT) rich DNA), like Hoechst dyes, forming a stable complex which fluoresces approximately 20 times greater than DAPI alone. Spectral Data: Excitation: λex 340 nm; Emission: λem 488 nm (only DAPI). Excitation: λex 360nm; Emission: λem 460nm (DAPI-DNA complex).
- Commonly used to stain DNA and chromosomes for fluorescent microscopy and flow cytometry applications.
- DAPI is often used as a counterstain, as its ultraviolet excitation (λex 360nm) and blue emission (λem 460nm) wavelengths separate it nicely from many popular primary fluorophores.
- Can be used on either fixed or live cells, although it passes through the membrane less efficiently in live cells and therefore the effectiveness of the stain is lower and demands higher concentrations to be used.
- Reversible inhibitor of S-adenosyl-L-methionine decarboxylase and KAO (diamine oxidase).
Product References
- A simple cytochemical technique for demonstration of DNA in cells infected with mycoplasmas and viruses: W.C. Russell, et al.; Nature 253, 461 (1975)
- DIPI and DAPI: fluorescence banding with only negliglible fading: W. Schnedl, et al.; Hum. Genet. 36, 167 (1977)
- An evaluation of DNA fluorochromes, staining techniques, and analysis for flow cytometry. I. Unperturbed cell populations: I.W. Taylor & B.K. Milthorpe; J. Histochem. Cytochem. 28, 1224 (1980)
- A comparative study of DAPI, DIPI, and Hoechst 33258 and 33342 as chromosomal DNA stains: F. Otto & K.C. Tsou; Stain Technol. 60, 7 (1985)
- Estimation of total DNA in crude extracts of plant leaf tissue using 4',6-diamidino-2-phenylindole (DAPI) fluorometry: L.S. Lee & H.M. Garnett; J. Biochem. Biophys. Methods 26, 249 (1993)
- 4',6-Diamidino 2-phenylindole is a new reversible inhibitor of diamine oxidase and S-adenosyl-L-methionine decarboxylase from mammalian tissues: C. Cubria, et al.; Comp. Biochem. Physiol. C. 105, 251 (1994)
- Flow cytometric detection of apoptosis: comparison of the assays of in situ DNA degradation and chromatin changes: M.A. Hotz, et al.; Cytometry 15, 237 (1994)
- DAPI: a DNA-specific fluorescent probe: J. Kapuscinski; Biotech. Histochem. 70, 220 (1995)
- The use of DAPI as a replacement for ethidium bromide in forensic DNA analysis: E. Buel & M. Schwartz; J. Forensic Sci. 40, 275 (1995)
- Visualizing chromatin and chromosomes in living cells: D. Zink, et al.; Methods: A Companion to Methods in Enzymology 29, 42 (2003)
- Propidium Iodide (PI) or DAPI Staining of Unfixed Solid Tissues for Flow Cytometry: L. Rodgers; CSH Protoc. 2006, pdb.prot4438 (2006)
- DAPI Staining of Drosophila Embryos: W.F. Rothwell & W. Sullivan; CSH Protoc. 2007, pdb.prot4834 (2007)
- Labeling nuclear DNA using DAPI: B. Chazotte; CSH Protoc. 2011, pdb.prot5556 (2011)
- An investigation of the photophysical properties of minor groove bound and intercalated DAPI through quantum-mechanical and spectroscopic tools: A. Biancardi, et al.; Phys. Chem. Chem. Phys. 15, 4596 (2013)
- DAPI staining and fluorescence microscopy techniques for phytoplasmas: N.M. Andrade & N.L. Arismendi; Methods Mol. Biol. 938, 115 (2013)
- The hazards of DAPI photoconversion: effects of dye, mounting media and fixative, and how to minimize the problem: M. Jez, et al.; Histochem. Cell Biol. 139, 195 (2013)
- Fluorescence lifetime imaging of DAPI-stained nuclei as a novel diagnostic tool for the detection and classification of B-cell chronic lymphocytic leukemia: G. Yahav, et al.; Cytometry A 89, 644 (2016)