Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
AdipoGen Life Sciences
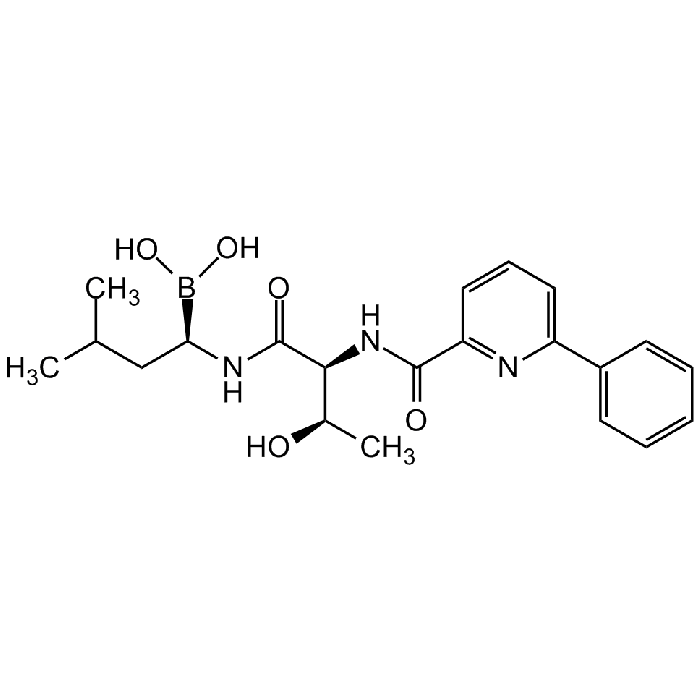
Delanzomib [CEP-18770]
As low as
40
CHF
CHF 40.00
In stock
Only %1 left
AG-CR1-3673-M0011 mgCHF 40.00
AG-CR1-3673-M0055 mgCHF 130.00
| Product Details | |
|---|---|
| Synonyms | NPH 007098; CT 47098; CIP 18770; [(1R)-1-[[(2S,3R)-3-Hydroxy-2-[(6-phenylpyridine-2-carbonyl)amino]butanoyl]amino]-3-methylbutyl]boronic acid |
| Product Type | Chemical |
| Properties | |
| Formula |
C21H28BN3O5 |
| MW | 413.3 |
| CAS | 847499-27-8 |
| Purity Chemicals | ≥95% (1H-NMR) |
| Appearance | White solid. |
| Solubility | Soluble in DMSO (20mg/ml) or ethanol (20mg/ml). Slightly soluble in chloroform or methanol. Poorly soluble in water (<1mg/ml). |
| Other Product Data |
Note: Warming and sonication may be required when dissolving the compound in the solvent of choice. Stock solutions are stable for at least 1 month when stored at -20°C. |
| InChi Key | SJFBTAPEPRWNKH-CCKFTAQKSA-N |
| Smiles | CC(C)C[C@@H](B(O)O)NC([C@H]([C@H](O)C)NC(C1=NC(C2=CC=CC=C2)=CC=C1)=O)=O |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice | Keep cool and dry. |
| Use/Stability | Stable for at least 2 years after receipt when stored at -20°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
- Potent, selective, reversible orally bioavailable proteasome inhibitor. Synthetic P2 threonine boronic acid.
- Targets the chymotrypsin-like β5 subunit of the constitutive 20S proteasome (IC50=3.8nM). Cross-reacts and inhibits the caspase-like/peptidyl-glutamyl peptide-hydrolyzing (PGPH) β1 subunit (IC50=~70nM).
- Displays similar potency for the chymotrypsin-like activity of the proteasome compared to bortezomib. Exhibits a favorable cytotoxicity profile toward normal human epithelial cells, bone marrow progenitors and bone marrow-derived stromal cells relative to bortezomib (Prod. No. AG-CR1-3602).
- Anticancer compound effective against multiple myeloma in vivo.
- In vitro, blocks the growth of representative human solid and hematological tumor cell lines (IC50s=5.6-34nM).
- Shown to down-modulate NF-κB, induce apoptosis, inhibit angiogenesis and M-CSF-RANKL-induced osteoclastogenesis.
Product References
- Discovery of a Potent, Selective, and Orally Active Proteasome Inhibitor for the Treatment of Cancer: B.D. Dorsey, et al.; J. Med. Chem. 51, 1068 (2008)
- CEP-18770: A novel, orally active proteasome inhibitor with a tumor-selective pharmacologic profile competitive with bortezomib: R. Piva, et al.; Blood 111, 2765 (2008)
- The proteasome inhibitor CEP-18770 enhances the anti-myeloma activity of bortezomib and melphalan: E. Sanchez, et al.; Br. J. Haematol. 148, 569 (2010)
- Novel, orally active, proteasome inhibitor, delanzomib (CEP-18770), ameliorates disease symptoms and glomerulonephritis in two preclinical mouse models of SLE: M.M. Seavey, et al.; Int. Immunopharmacol. 12, 257 (2012)
- Probing the specificity and activity profiles of the proteasome inhibitors bortezomib and delanzomib: C.R. Berkers, et al.; Mol. Pharm. 9, 1126 (2012)
- Molecular mechanisms of acquired proteasome inhibitor resistance: A.J. Kale & B.S. Moore; J. Med. Chem. 55, 10317 (2012)
- A first in human phase I study of the proteasome inhibitor CEP-18770 in patients with advanced solid tumours and multiple myeloma: E. Gallerani, et al.; Eur. J. Cancer 49, 290 (2013)
- Proteasome inhibitors in the treatment of multiple myeloma: A. McBride & P.Y. Ryan; Expert Rev. Anticancer Ther. 13, 339 (2013) (Review)
- Phase I/II study of the novel proteasome inhibitor delanzomib (CEP-18770) for relapsed and refractory multiple myeloma: D.T. Vogl, et al.; Leuk. Lymphoma 58, 1872 (2017)






![VR23 [Proteasome Inhibitor]](https://adipogen.com/media/catalog/product/cache/60eb5af712bc93baae8d55513bd31b01/a/g/ag-cr1-3676.png)
![PI-1840 [Proteasome Inhibitor]](https://adipogen.com/media/catalog/product/cache/60eb5af712bc93baae8d55513bd31b01/a/g/ag-cr1-3675.png)
![Oprozomib [ONX 0912]](https://adipogen.com/media/catalog/product/cache/60eb5af712bc93baae8d55513bd31b01/a/g/ag-cr1-3672.png)
![Ixazomib citrate [MLN9708]](https://adipogen.com/media/catalog/product/cache/60eb5af712bc93baae8d55513bd31b01/a/g/ag-cr1-3671.png)
![Ixazomib [MLN2238]](https://adipogen.com/media/catalog/product/cache/60eb5af712bc93baae8d55513bd31b01/a/g/ag-cr1-3670.png)
![Carfilzomib [PR-171]](https://adipogen.com/media/catalog/product/cache/60eb5af712bc93baae8d55513bd31b01/a/g/ag-cr1-3669.png)



