Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
BioViotica
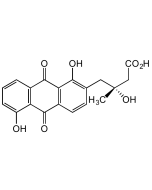
Fridamycin A
As low as
110
CHF
CHF 110.00
In stock
Only %1 left
BVT-0469-M0011 mgCHF 110.00
BVT-0469-M0055 mgCHF 325.00

| Product Details | |
|---|---|
| Synonyms | Vineomycinone B2 |
| Product Type | Chemical |
| Properties | |
| Formula |
C25H26O10 |
| MW | 486.5 |
| CAS | 30270-05-4 |
| Source/Host Chemicals | Isolated from Streptomyces parvulus. |
| Purity Chemicals | ≥97% (HPLC, NMR) |
| Appearance | Yellow to orange solid. |
| Solubility | Soluble in DMSO. Sparingly soluble in methanol or acetone. |
| Identity | Determined by 1H-NMR and MS. |
| Declaration | Manufactured by BioViotica. |
| InChi Key | LKLNNJGYAYDANM-VRSSYPSJSA-N |
| Smiles | [H][C@@]1(C[C@@H](O)[C@H](O)[C@@H](C)O1)C1=C(O)C2=C(C=C1)C(=O)C1=C(C=CC(C[C@@](C)(O)CC(O)=O)=C1O)C2=O |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice | Protect from light. |
| Use/Stability |
Stable for at least 1 year after receipt when stored at -20°C. Store solutions at -20°C in the dark. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
- C-glycosidic angucyclinone derivative.
- Antibiotic.
- Antitumor agent.
- Antibacterial agent.
- Isomer of fridamycin B (Prod. No. BVT-0470).
Product References
- The structure of vineomycin B2: N. Imamura, et al.; J. Antibiot. 34, 1517 (1981)
- Fridamycine, Anthracyclin-analoge Antibiotica: P. Knicke; Ph. D. Thesis Univ. Goettingen (1984)
- Angucycline group antibiotics: J. Rohr & R. Thiericke; Nat. Prod. Rep. 9, 103 (1992)
- Synthetic anthracyclines from anthraquinones: R. Cambie, et al.; Austr. J. Chem. 45, 483 (1992)
- C-Aryl glycosides via tandem intramolecular benzyne-furan cycloadditions. Total synthesis of vineomycine B2 methyl ester: C.-L. Chen, et al.; JACS 128, 13696 (2006)
- Convergent de novo synthesis of vineomycinione B2 methyl ester: Q. Chen, et al.; Chem. Comm. 49, 6806 (2013)
- Total Synthesis of vineomycin B2: S. Kusumi, et al.; JACS 135, 15909 (2013)
- Angucycline antibiotics and its derivatives from marine-derived actinomycete streptomyces sp. A6H: Z. Hu, et al.; Nat. Prod. Res. 30, 2551 (2016)