Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
BioViotica
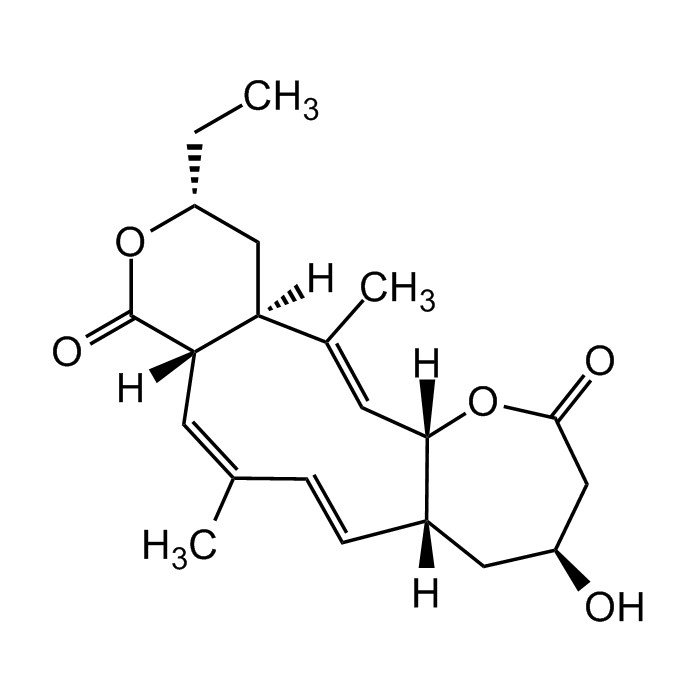
Collinolactone
As low as
190
CHF
CHF 190.00
In stock
Only %1 left
BVT-0480-C250250 µgCHF 190.00
BVT-0480-M0011 mgCHF 575.00
| Product Details | |
|---|---|
| Synonyms | Rhizolutin |
| Product Type | Chemical |
| Properties | |
| Formula |
C21H28O5 |
| MW | 360.45 |
| CAS | 2733581-46-7 |
| Source/Host Chemicals | Isolated from Streptomyces sp. |
| Purity Chemicals | ≥98% (HPLC) |
| Appearance | White to off-white solid. |
| Solubility | Soluble in DMSO, pyridine, toluene, benzene or acetonitrile. Insoluble in water. We recommend to avoid solutions in methanol. Protect from pH-values below pH 4.0 and above pH 8.0. |
| Identity | Determined by coupled HPLC-UV-high resolution mass spectrometry (ESI(+)-TOF), 1H-NMR and 13C-NMR spectroscopy. |
| Declaration | Manufactured by BioViotica. |
| Other Product Data |
Note: The NMR-spectra shows a mixture of two conformers (ratio 4:1) due to the rotating of the propenyl unit resulting in a syn- and anti-rotamer. |
| InChi Key | GSVYLEFSSDOSSC-QHEHVBGPSA-N |
| Smiles | O=C1O[C@H](CC)C[C@]2([H])[C@]1([H])/C=C(C)\C=C\[C@](C[C@H](O)CC(O3)=O)([H])[C@]3([H])/C=C2\C |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice |
Keep cool and dry. Protect from light when in solution. |
| Use/Stability |
Stable for at least 1 year after receipt when stored at -20°C. Store solutions at -20°C in the dark. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
- Collinolactone has neuroprotective and anti-neurodegenerative effects by reducing intracellular oxidative stress. It protects against glutamate-induced oxidative stress in a dose-dependent manner.
- Collinolactone is a potential anti-Alzheimer agent with Aβ-disaggregating activity. It showed dissociative effect on Aβ aggregates and tau tangles in vitro, with significantly reduced apoptosis and inflammation in neuronal and glial cells. In vivo studies on APP/PS1 double transgenic mice showed substantially dissociated hippocampal plaques.
- A set of semisynthetic collinolactone analogs (but not collinolactone) has shown inhibitory effects on a L929 cell line.
Product References
- Big effects from small changes: possible ways to explore nature's chemical diversity: H.B. Bode, et al.; Chembiochem 3, 619 (2002)
- Rhizolutin, a novel 7/10/6-tricyclic dilactone, dissociates misfolded protein aggregates and reduces apoptosis/inflammation associated with Alzheimer's Disease: Y. Kwon, et al.; Angew. Chem. Int. Ed. 59, 22994 (2020)
- Structure of cyclodecatriene collinolactone, its biosynthesis, and semisynthetic analogues: effects of monoastral phenotype and protection from intracellular oxidative stress: J.C. Schmid, et al.; Angew. Chem. Int. Ed. 60, 23212 (2021)