Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
Chemodex
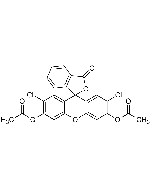
Carboxy-DCFDA
| Product Details | |
|---|---|
| Synonyms | 5(6)-Carboxy-2′,7′-dichlorofluorescein diacetate |
| Product Type | Chemical |
| Properties | |
| Formula |
C25H14Cl2O9 |
| MW | 529.28 |
| CAS | 127770-45-0 |
| Source/Host Chemicals | Synthetic. |
| Purity Chemicals | ≥85% (HPCE) |
| Appearance | Off-white to light orange powder. |
| Solubility | Soluble in DMSO, DMF or methanol. |
| Identity | Determined by 1H-NMR. |
| Declaration | Manufactured by Chemodex. |
| Other Product Data |
Click here for Original Manufacturer Product Datasheet |
| Smiles | CC(O)=O.CC(=O)OC1=CC2=C(C=C1Cl)C1(OC(=O)C3=C1C=CC=C3)C1=CC(Cl)=C(OC(C)=O)C=C1O2 |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | +4°C |
| Handling Advice | Protect from light and moisture. |
| Use/Stability | Stable for at least 2 years after receipt when stored at +4°C. |
| Documents | |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Non-activated, amine-reactive fluorescent probe. Mixture of 5/6-isomers of CDCFDA that can be used in biomolecules conjugation. 5(6)-CDCFDA is an amine–reactive fluorescein diacetate (FDA) derivative used to prepare a variety of FDA conjugates. 5(6)-CDCFDA is a non-fluorescent molecule that diffuse into cells and is hydrolyzed by intracellular non-specific esterases, yielding a fluorescent probe. The probe accumulates exclusively in viable cells with intact cell membranes. 5(6)-CDCFDA does not stain dead cells. 5(6)-CDCFDA labeling can be used for monitoring cells by flow cytometry or fluorescence microscopy. The probe is also frequently used to monitor reactive oxygen species (ROS) generation in live cells and intracellular pH levels. Spectral data: λex=504nm, λem=529nm.
(1) J.P. Kehrer & T. Paraidathathu; Free Rad. Res. Commun. 16, 217 (1992) | (2) Q.P. Lloyd, et al.; J. Biol. Chem. 270, 22445 (1995) | (3) A.L. Inselman, et al.; Curr. Microbiol. 38, 295 (1999) | (4) M.J. Zamek-Gliszczynski, et al.; J. Pharmacol. Exp. Therap. 304, 801 (2003) | (5) L. Li, et al.; Free Radic. Res. 42, 354 (2008) | (6) M. Medhora, et al.; Am. J. Physiol. Lung Cell Mol. Physiol. 294, L902 (2008) | (7) S. Siissalo, et al.; Eur. J. Pharm. Biopharm. 71, 332 (2009) | (8) A.I. Pogue, et al.; Int. J. Mol. Sci. 13, 9615 (2012) | (9) J. Roh, et al.; Redox Biol. 11, 254 (2016) | J.L. Roh, et al.; Cancer Lett. 381, 96 (2016)