Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
Chimerigen
IL-35 (mouse):Fc (human) (rec.)
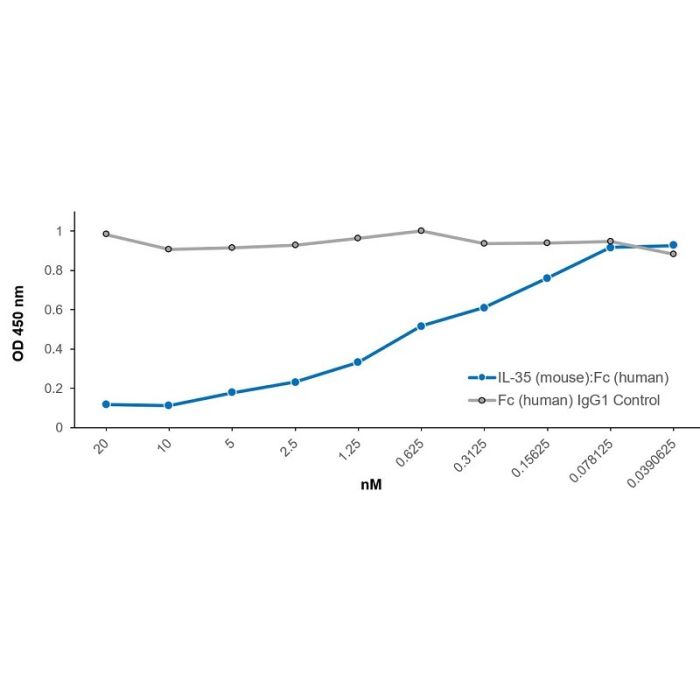
Method: 5x10^4 Raw 264.7 cells are treated with IL-35 (mouse):Fc (human) (rec.) (Prod. No. CHI-MF-11135) or a control, Fc (human) IgG1 Control (rec.) (Prod. No. AG-35B-0007) for 16 hours at different concentrations as indicated on the figure. Then LPS (1µg/ml; Prod. No. IAX-100-008-C500) is added for another 24 hours. Supernatants are collected and mouse IL-6 is measured using an IL-6 (mouse) ELISA Kit. OD 450nm values indicating presence of mouse IL-6 are shown.
| Product Details | |
|---|---|
| Synonyms | Interleukin-35 |
| Product Type | Protein |
| Properties | |
| Source/Host | CHO cells |
| Sequence |
A soluble dimeric fusion protein consisting of the extracellular domain of mouse IL12a subunit (aa 23-215) is fused to the Fc region of human IgG1, and the mouse Ebi3 subunit (aa 23-228) linked to IL12a by disulfide bonds. |
| Crossreactivity | Mouse |
| Biological Activity |
Bioactivity was measured in a cell proliferation assay of Con A activated mouse splenocytes. |
| MW | ~30-60kDa |
| Purity | ≥95% (SDS-PAGE) |
| Endotoxin Content | <1EU/mg protein (LAL test; Lonza). |
| Reconstitution | Reconstitute at 100μg/ml in sterile PBS. |
| Formulation | Lyophilized from 0.2μm-filtered solution in PBS. |
| Protein Negative Control | |
| Other Product Data |
NCBI reference NP_056581.1: Ebi3 subunit (mouse) |
| Declaration | Manufactured by Chimerigen. |
| Shipping and Handling | |
| Shipping | BLUE ICE |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice |
Avoid freeze/thaw cycles. Centrifuge lyophilized vial before opening and reconstitution. |
| Use/Stability |
Stable for at least 1 year after receipt when stored at -20°C. Working aliquots are stable for up to 3 months when stored at -20°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Interleukin-35 (IL-35) is a novel IL-12 family cytokine produced by regulatory T cells (Treg) but not by resting or activated effector T cells (Teff). IL-35 is a heterodimeric protein composed of EBI3 (Epstein-Barr-Virus-induced gene 3) and IL-12a (p35). EBI3 is a downstream target of Foxp3, a transcription factor required for Treg-cell development and function, and thus Treg-cell restriction of IL35 occurs. Regulatory T cells are essential for maintaining self tolerance and preventing autoimmunity, and IL-35 is identified as a molecule that mediates the immune suppression function of Treg-cell. As an inhibitory cytokine, IL-35 induces proliferation of Treg-cell populations but suppresses Th17 cell development. Studies in mice show the absence of either IL-35 chain from Treg-cell reduces the cells' ability to suppress inflammation using an experimental model for inflammatory bowel disease. IL-35 is suggested as a potential target of immunotherapy. Recently, insufficient IL-35 levels were shown to play a pivotal role in the development of type 1 diabetes (T1D) and autoimmune diseases.
- Interleukin-35 administration counteracts established murine type 1 diabetes - possible involvement of regulatory T cells: K. Singh, et al.; Sci. Rep. 5, ID12633 (2015)
- Cytokine modulation by IL-35 in mice with allergic rhinitis: M. Yokota, et al.; Am. J. Rhinol. Allergy 29, 251 (2015)
- Remission of systemic lupus erythematosus disease activity with regulatory cytokine interleukin (IL)-35 in Murphy Roths Large (MRL)/lpr mice: Z. Cai, et al.; Clin. Exp. Immunol. 181, 253 (2015)
- Intranasal administration of IL-35 inhibits allergic responses and symptoms in mice with allergic rhinitis: M. Suzuki, et al.; Allergol. Int. 66, 351 (2017)
- IL-35 suppresses lipopolysaccharide-induced airway eosinophilia in EBI3-deficient mice: K. Kanai, et al.; J. Immunol. 198, 119 (2017)
- The unknown aspect of BAFF: Inducing IL-35 production by a CD5+CD1dhiFcγRIIbhi regulatory B-Cell subset in lupus: Y. Zhang, et al.; J. Invest. Dermatol. 137, 2532 (2017)
- IL-35 induces N2 phenotype of neutrophils to promote tumor growth: J.-M. Zou, et al.; Oncotarget 8, 33501 (2017)
- Regulatory T cells and their derived cytokine, interleukin-35, reduce pain in experimental autoimmune encephalomyelitis: S.S. Duffy, et al.; J. Neurosci. 39, 2326 (2019)
- Elevated interleukin-35 suppresses liver inflammation by regulation of T helper 17 cells in acute hepatitis B virus infection: D.K. Teng, et al.; Int. Immunopharm. 70, 252 (2019)
- Interleukin-33 activates regulatory T cells to suppress innate γδ T cell responses in the lung: L.D. Fasutino, et al. Nat. Immunol. 21, 1371 (2020)
- Elevation in the counts of IL-35-producing B cells infiltrating into lung tissue in mycobacterial infection is associated with the downregulation of Th1/Th17 and upregulation of Foxp3 + Treg: C. Chen, et al.; Sci. Rep. 10, 13212 (2020)
- B cell–Derived IL35 Drives STAT3-Dependent CD8+ T-cell Exclusion in Pancreatic Cancer: B. Mirlekar, et al.; Cancer Immunol. Res. 8, 292 (2020)
- Interleukin 35 delays hindlimb ischemia-induced angiogenesis through regulating ROS-extracellular matrix but spares later regenerative angiogenesis: H. Fu, et al.; Front. Immunol. 11, 595813 (2020)
- IL-35 promotes CD4+Foxp3+ Tregs and inhibits atherosclerosis via maintaining CCR5-amplified Treg-suppressive mechanisms: Y. Shao, et al.; JCI Insight 6, e152511 (2021)
- IL-35 Stabilizes Treg Phenotype to Protect Cardiac Allografts in Mice: A. Huang, et al.; Transplantation 108, 161 (2024)






