Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
AdipoGen Life Sciences
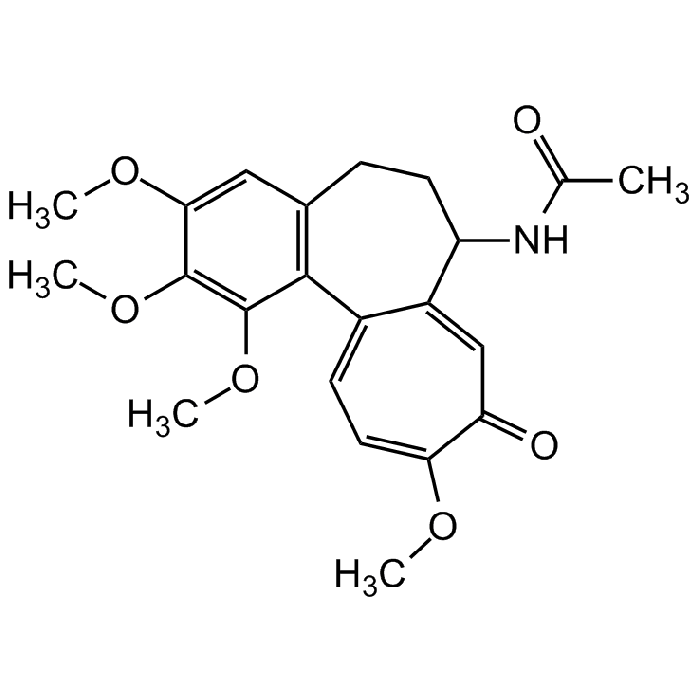
Colchicine
As low as
80
CHF
CHF 80.00
In stock
Only %1 left
AG-CN2-0048-M500500 mgCHF 80.00
AG-CN2-0048-G0011 gCHF 140.00
| Product Details | |
|---|---|
| Synonyms | NSC 757; EINECS 200-598-5 |
| Product Type | Chemical |
| Properties | |
| Formula |
C22H25NO6 |
| MW | 399.4 |
| Merck Index | 14: 2471 |
| CAS | 64-86-8 |
| RTECS | GH0700000 |
| Source/Host Chemicals | Isolated from Colchicum autumnale. |
| Purity Chemicals | ≥97% |
| Appearance | White to off-white powder. |
| Solubility | Soluble in 100% ethanol (>100mg/ml), DMSO (>100mg/ml), water or DMF. |
| Identity | Determined by 1H-NMR. |
| InChi Key | IAKHMKGGTNLKSZ-UHFFFAOYSA-N |
| Smiles | COC1=CC2=C(C(OC)=C1OC)C1=CC=C(OC)C(=O)C=C1C(CC2)NC(C)=O |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice | Protect from light and moisture. |
| Use/Stability | Stable for at least 2 years after receipt when stored at -20°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
- Anti-cancer compound [5].
- Microtubule assembly inhibitor. Depolymerizes microtubules and limits microtubule formation (inactivates spindle fibre formation) [1, 2, 5, 6].
- Inhibits mitosis during cell division at metaphase by inhibiting spindle formation [6].
- Anti-inflammatory compound [7, 9]. Suppresses monosodium urate crystal-induced NLRP3/NALP3 inflammasome-driven caspase-1 activation, IL-1β processing and release, and L-selectin expression on neutrophils at micromolar concentrations. Blocks the release of a crystal-derived chemotactic factor from neutrophil lysosomes, blocks neutrophil adhesion to, and inhibits monosodium urate crystal-induced production of superoxide anions from neutrophils at nanomolar concentrations [7].
- Drug used in treatment of gout, familial Mediterranean fever, pericarditis and Behect's disease. Investigated for its anti-cancer activity [5, 8, 9]. It has a narrow therapeutic index with no clear-cut distinction between nontoxic, toxic and lethal doses, causing substantial confusion among clinicians [8].
- Apoptosis inducer in a variety of normal and tumor cell lines [3, 4].
- Inhibitor of autophagosome-lysosome fusion.
- Inhibits acetylated a-tubulin mediated dynein dependent transport of mitochondria and subsequent apposition of ASC on mitochondria to NLRP3 on the endoplasmic reticulum in vitro and in vivo.
Product References
- Interactions of colchicine with tubulin: S.B. Hastie; Pharmacol. Ther. 51, 377 (1991) (Review)
- Analysis of the colchicine-binding site of beta-tubulin: R.G. Burns; FEBS Lett. 297, 205 (1992) (Review)
- Colchicine induces apoptosis in cerebellar granule cells: E. Bonfoco, et al.; Exp. Cell. Res. 218, 189 (1995)
- Sustained JNK activation induces endothelial apoptosis: studies with colchicine and shear stress: Y.L. Hu, et al.; Am. J. Physiol. 277, H1593 (1999)
- Microtubules as a target for anticancer drugs: M.A. Jordan & L. Wilson; Nat. Rev. Cancer 4, 253 (2004)
- Anti-mitotic activity of colchicine and the structural basis for its interaction with tubulin: B. Bhattacharyya, et al.; Med. Res. Rev. 28, 155 (2008) (Review)
- Colchicine: its mechanism of action and efficacy in crystal-induced inflammation: G. Nuki; Curr. Rheumatol. Rep. 10, 218 (2008) (Review)
- Colchicine poisoning: the dark side of an ancient drug: Y. Finkelstein, et al.; Clin. Toxicol. (Phila). 48, 407 (2010) (Review)
- Colchicine for the treatment of gout: P. Richette & T. Bardin; Expert Opin. Pharmacother. 11, 2933 (2010) (Review)
- Microtubule-driven spatial arrangement of mitochondria promotes activation of the NLRP3 inflammasome: T. Misawa, et al.; Nat. Immunol. 14, 454 (2013)
- Nuclear receptor coactivator 6 is a critical regulator of NLRP3 inflammasome activation and gouty arthritis: K-G. Lee, et al.; Cell. Mol. Immunol. ahead of print (2024)