Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
AdipoGen Life Sciences
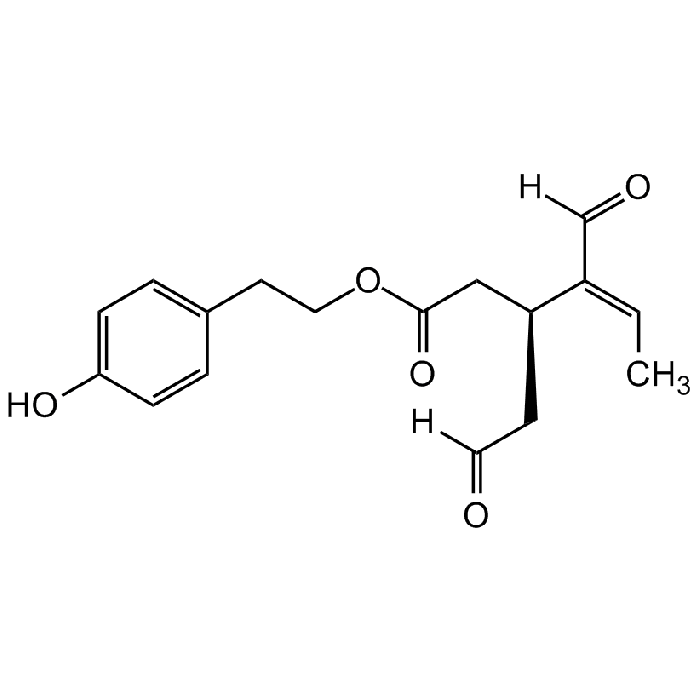
Oleocanthal
As low as
240
CHF
CHF 240.00
In stock
Only %1 left
AG-CN2-0537-M0055 mgCHF 240.00
| Product Details | |
|---|---|
| Synonyms | (-)-Oleocanthal; 2-(4-Hydroxyphenyl)ethyl (3S,4E)-4-formyl-3-(2-oxoethyl)hex-4-enoate; Deacetoxy Ligstroside Aglycon; p-HPEA-EDA |
| Product Type | Chemical |
| Properties | |
| Formula |
C17H20O5 |
| MW | 304.3 |
| CAS | 289030-99-5 |
| Source/Host Chemicals | Isolated from Olea europaea. |
| Purity Chemicals | ≥97% (HPLC) |
| Appearance | Amber oil. |
| Solubility | Soluble in DMSO, dichloromethane or acetonitrile. Slightly soluble in methanol, ethanol or water (0.1mg/ml). |
| Identity | Determined by 1H-NMR. |
| Other Product Data |
Note: Shown to react spontaneously with water or methanol to give mixtures of hemiacetals or acetals (see Karkoula et al 2012). |
| InChi Key | VPOVFCBNUOUZGG-VAKDEWRISA-N |
| Smiles | OC1=CC=C(CCOC(C[C@H](CC([H])=O)/C(C([H])=O)=C\C)=O)C=C1 |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice | Keep cool and dry. |
| Use/Stability | Stable for at least 2 years after receipt when stored at -20°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
- Oleocanthal is a naturally occurring amphiphilic secoiridoid from olive oil, with potent anti-inflammatory, antioxidant, anticancer and neuroprotective properties.
- Similar to classical non-steroidal anti-inflammatory drugs, it is a non-selective inhibitor of cyclooxygenase (COX-1, COX-2) enzymes in the prostaglandin biosynthesis. It also has been shown to inhibit the NLRP3 inflammasome.
- Inhibits proliferation, migration and invasion of various cancer cells while leaving healthy cells unharmed. It shows selective cytotoxicity, inducing lysosomal membrane permeabilization in cancer cells. It inhibits the enzymatic activity of mammalian target of rapamycin (mTOR), and the tyrosine kinase receptor c-Met (responsible for proliferation of many cell types) and Hsp90, consequently leading to cell cycle arrest during G1 phase.
- The neuroprotective activity includes the reduction of β-amyloid protein accumulation via up-regulation of P-glycoprotein and LRP1, altering the fibrillization of tau proteins, key factors at the basis of neurodegenerative diseases and reducing oxidative stress.
- Activator of the TRPA1 ion channel, responsible for the burning sensation that occurs in the back of the throat when consuming such oil.
- Shows antimicrobial activities.
Product References
- Simple and hydrolyzable compounds in virgin olive oil. 3. Spectroscopic characterizations of the secoiridoid derivatives: G. Montedoro, et al.; J. Agric. Food Chem. 41, 2228 (1993)
- Phytochemistry: ibuprofen-like activity in extra-virgin olive oil: G.K. Beauchamp, et al.; Nature 437, 7055 (2005)
- Synthesis and assignment of absolute configuration of (-)-oleocanthal: a potent, naturally occurring non-steroidal anti-inflammatory and anti-oxidant agent derived from extra virgin olive oils: A.B. Smith, et al.; Org. Lett. 7, 5075 (2005)
- Comparison of the concentrations of phenolic compounds in olive oils and other plant oils: correlation with antimicrobial activity: E. Medina, et al.; J. Agric. Food Chem. 54, 4954 (2006)
- Sensory characterization of the irritant properties of oleocanthal, a natural anti-inflammatory agent in extra virgin olive oils: S. Cicerale, et al.; Chem. Senses 34, 333 (2009)
- (-)-Oleocanthal as a c-Met inhibitor for the control of metastatic breast and prostate cancers: A.Y. Elnagar, et al.; Planta Med. 77, 1013 (2011)
- Olive-oil-derived oleocanthal enhances β-amyloid clearance as a potential neuroprotective mechanism against Alzheimer's disease: in vitro and in vivo studies: A.H. Abuznait, et al.; ACS Chem. Neurosci. 4, 973 (2013)
- Modulation of tau protein fibrillization by oleocanthal: M.C. Monti, et al.; J. Nat. Prod. 75, 1584 (2012)
- Direct Measurement of Oleocanthal and Oleacein Levels in Olive Oil by Quantitative 1H NMR. Establishment of a New Index for the Characterization of Extra Virgin Olive Oils: E. Karkoula, et al.; J. Agric. Food Chem. 60, 11696 (2012)
- Chemical proteomics-driven discovery of oleocanthal as an Hsp90 inhibitor: L. Margarucci, et al.; Chem. Commun. 49, 5844 (2013)
- Olive phenolics as c-Met inhibitors: (-)-Oleocanthal attenuates cell proliferation, invasiveness, and tumor growth in breast cancer models: M.R. Akl, et al.; PLoS One 9, e97622 (2014)
- New drugs from ancient natural foods. Oleocanthal, the natural occurring spicy compound of olive oil: a brief history: M. Scotece, et al.; Drug Discov. Today 20, 406 (2015) (Review)
- Olive Oil-derived Oleocanthal as Potent Inhibitor of Mammalian Target of Rapamycin: Biological Evaluation and Molecular Modeling Studies: M.A. Khanfar, et al.; Phytother. Res. 29, 1776 (2015)
- Current Disease-Targets for Oleocanthal as Promising Natural Therapeutic Agent: A. Segura-Carretero & J.A. Curiel; Int. J. Mol. Sci. 19, E2899 (2018) (Review)
- New neuroprotective perspectives in fighting oxidative stress and improving cellular energy metabolism by oleocanthal: C. Angeloni, et al.; Neural. Regen. Res. 14, 1217 (2019)
- Oleocanthal-Rich Extra-Virgin Olive Oil Restores the Blood-Brain Barrier Function through NLRP3 Inflammasome Inhibition Simultaneously with Autophagy Induction in TgSwDI Mice: S.B. Al Rihani, et al.; ACS Chem. Neurosci. 10, 3543 (2019)
- (-)-Oleocanthal and (-)-oleocanthal-rich olive oils induce lysosomal membrane permeabilization in cancer cells: L. Goren, et al.; PLoS One 14, e0216024 (2019)