Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
BioViotica
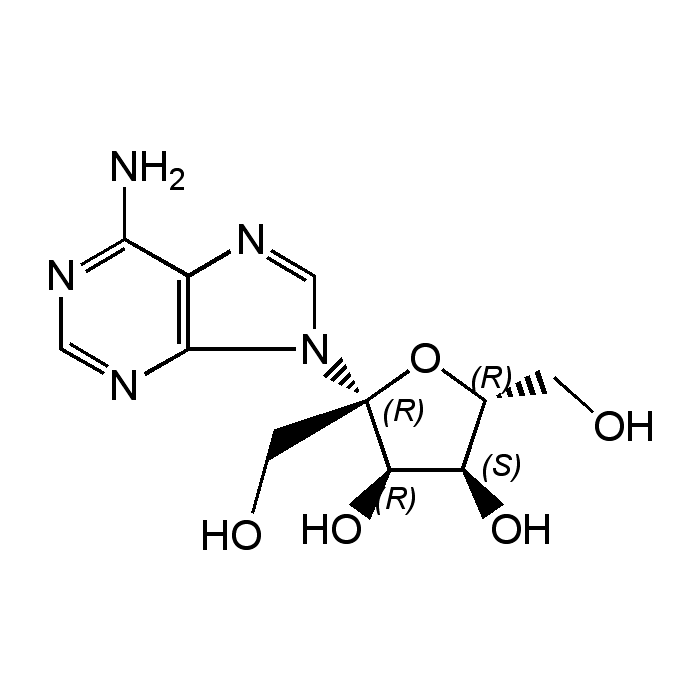
Psicofuranine
As low as
95
CHF
CHF 95.00
In stock
Only %1 left
BVT-0284-M0011 mgCHF 95.00
BVT-0284-M0055 mgCHF 380.00
| Product Details | |
|---|---|
| Synonyms | Angustmycin C; U-9586; 9-β-D-Psicofuranosyl-9H-purin-6-amine |
| Product Type | Chemical |
| Properties | |
| Formula |
C11H15N5O5 |
| MW | 297.3 |
| Merck Index | 14: 7923 |
| CAS | 1874-54-0 |
| Source/Host Chemicals | Isolated from Streptomyces sp. S 2113. |
| Purity Chemicals | ≥98% (HPLC, TLC) |
| Appearance | White to off-white solid. |
| Solubility | Soluble in DMSO; poorly soluble in methanol. |
| Identity | Determined by 1H-NMR. |
| Declaration | Manufactured by BioViotica. |
| InChi Key | BNZYRKVSCLSXSJ-IOSLPCCCSA-N |
| Smiles | NC1=NC=NC2=C1N=CN2[C@]1(CO)O[C@H](CO)[C@@H](O)[C@H]1O |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice | Avoid freeze/thaw cycles. |
| Use/Stability |
Stable for at least 1 year after receipt when stored at -20°C. After reconstitution protect from light at -20°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
- Nucleoside antibiotic.
- Antitumor compound.
- Xanthosine monophosphate (XMP) aminase inhibitor.
- Antimetabolite of the purine biosynthesis.
Product References
- Studies on angustmycins VIII. The structure of angustmycin C: H. Yüntsen; J. Antibiot. 11A, 244 (1958)
- Psicofuranine I. Discovery, isolation, and properties: T.E. Eble, et al.; Antibiot. Chemother. 9, 419 (1959)
- Psicofuranine. VI. Antitumor and toxicopathological studies: J.S. Evans & J.E. Gray; Antibiot. Chemother. 9, 675 (1959)
- Mechanism of action of psicofuranine: L.J. Hanka; J. Bacteriol. 80, 30 (1960)
- Inhibition of parental and mutant xanthosine 5'-phosphate aminases by psicofuranine: S. Udaka & H.S. Moyed; J. Biol. Chem. 238, 2797 (1963)
- Formation of an adenylxanthosinemonophosphate intermediate by xanthosine 5'-phosphate aminase and its inhibition by psicofuranine: T.T. Fukuyama; J. Biol. Chem. 241, 4745 (1966)
- Nucleoside antibiotics: structure, biological activity, and biosynthesis: K. Isono; J. Antibiot. 41, 1711 (1988), (Review)
- Role of purine biosynthetic intermediates in response to folate stress in Escherichia coli: C.E. Rohlman & R.G. Matthews; J. Bacteriol. 172, 7200 (1990)
- Plasmodium falciparum: isolation and characterisation of a gene encoding protozoan GMP synthase: G.A. McConkey; Exp. Parasitol. 94, 23 (2000)