Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
BioViotica
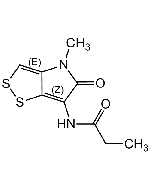
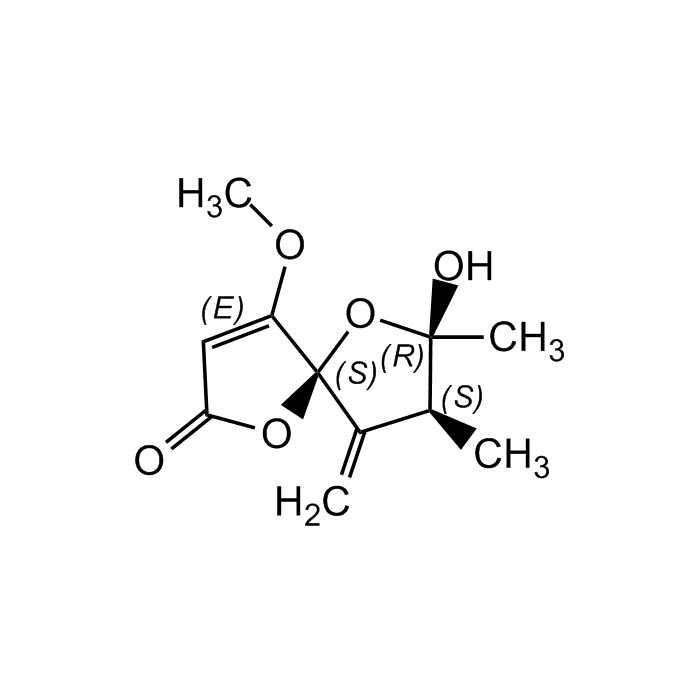
Papyracillic acid A
As low as
170
CHF
CHF 170.00
In stock
Only %1 left
BVT-0308-C500500 µgCHF 170.00
BVT-0308-M0011 mgCHF 265.00
| Product Details | |
|---|---|
| Synonyms | (5S,7R,8S)-7-Hydroxy-4-methoxy-7,8-dimethyl-9-methylene-1,6-dioxaspiro[4.4]non-3-en-2-one |
| Product Type | Chemical |
| Properties | |
| Formula |
C11H14O5 |
| MW | 226.2 |
| CAS | 960148-59-8 |
| Source/Host Chemicals | Isolated from Lachnum papyraceum and a Microsphaeropsis sp. |
| Purity Chemicals | ≥98% (HPLC) |
| Appearance | Off-white to beige solid. |
| Solubility | Soluble in dichloromethane or chloroform. |
| Identity | Determined by 1H-NMR. |
| Declaration | Manufactured by BioViotica. |
| InChi Key | UEYMIBGYKZFDMD-MQOMDTIOSA-N |
| Smiles | COC1=CC(=O)O[C@]11O[C@@](C)(O)[C@@H](C)C1=C |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Use/Stability |
Stable for at least 1 year after receipt when stored at -20°C. After reconstitution protect from light at -20°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
- Antibiotic. Exists in an equilibrium of diastereomers.
- Antibacterial and antifungal.
- Shows strong mosquito biting deterrent activity.
- Phytotoxin. Mycoherbicide.
Product References
- Papyracillic acid, a new penicillic acid analogue from the ascomycete Lachnum papyraceum: R. Shan, et al.; Tetrahedron 52, 10249 (1996)
- The reactivity of the fungal toxin papyracillic acid: R. Shan, et al.; Tetrahedron 53, 6209 (1997)
- Metabolic products of the endophytic fungus Microsphaeropsis sp. from Larixdecidua: J. Dai, et al.; Eur. J. Org. Chem. 2007, 4845 (2007)
- Papyracillic acid, a phytotoxic 1,6-dioxaspiro[4,4]nonene produced by Ascochyta agropyrina Var. nana, a potential mycoherbicide for Elytrigia repens biocontrol: A. Evidente, et al.; J. Agric. Food Chem. 57, 11168 (2009)
- Cyclopaldic acid, seiridin, and sphaeropsidin A as fungal phytotoxins, and larvicidal and biting deterrents against Aedes aegypti (Diptera: Culicidae): structure-activity relationships: A: Cimmino, et al.; Chem. Biodivers. 10, 1239 (2013)
- Fungal metabolites antagonists towards plant pests and human pathogens: Structure-activity relationship studies: M. Masi, et al.; Molecules 23, 834 (2018) (Review)