Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
Chemodex
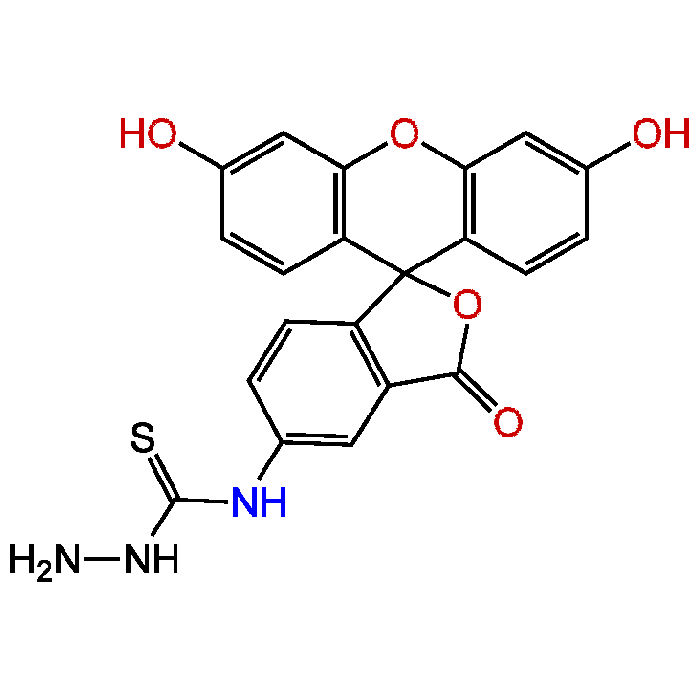
Fluorescein-5-thiosemicarbazide
| Product Details | |
|---|---|
| Synonyms | 5-FTSC; FTSC, FTZ; N-(3',6'-Dihydroxy-3-oxospiro[isobenzofuran-1(3H),9'-[9H]xanthen]-5-yl)-hydrazinecarbothioamide |
| Product Type | Chemical |
| Properties | |
| Formula | C21H15N3O5S |
| MW | 421.43 |
| CAS | 76863-28-0 |
| Source/Host Chemicals | Synthetic. |
| Purity Chemicals | ≥97% (NMR) |
| Appearance | Orange to brown powder. |
| Solubility | Soluble in water or DMF. |
| Identity | Determined by 1H-NMR. |
| Declaration | Manufactured by Chemodex. |
| Other Product Data |
Click here for Original Manufacturer Product Datasheet |
| InChi Key | OIMMDIXLTJAVTF-UHFFFAOYSA-N |
| Smiles | NNC(=S)NC1=CC2=C(C=C1)C1(OC2=O)C2=C(OC3=C1C=CC(O)=C3)C=C(O)C=C2 |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice | Protect from light and moisture. |
| Use/Stability | Stable for at least 2 years after receipt when stored at -20°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Hydrazide derivative of fluorescein. An amine-containing cell impermeant fluorescent probe that can be reversibly coupled to aldehydes and ketones on diverse molecules. This forms a Schiff base - which can be further reduced to generate stable amine derivatives by treatment with reducing agents such as sodium borohydride (NaBH4) or sodium cyanoborohydride (NaCNH3). Carboxylic acids of proteins and other water-soluble biopolymers can also be coupled to this fluorescein derivative in aqueous solution using water-soluble carbodiimides such as EDAC. Fluorescent tag for labeling of cell-surface functional groups (glycophorins) and many other diverse molecules, including DNA, RNA, polysaccharides, sialylated glycoproteins, carbonylated proteins, carbonyl derivatives, and N-acetylneuraminic acid. Useful for determining protein and peptide topology on the cell surface. This probe has been used for directly imaging of mucin-type O-linked glycoproteins within living cells, for imaging polysaccharides transported in living cells and for fluorescence labeling of short RNA by oxidation at the 3′-End. Spectral data: λex=492nm; λem=516nm (in 0.1 M Tris pH9.0).
(1) D.H. Atha, et al.; Biochim. Biophys. Acta 785, 1 (1984) | (2) B. Ahn, et al.; Anal. Biochem. 161, 245 (1987) | (3) W.A. Duijndam, et al.; J. Immunol. Methods 109, 289 (1988) | (4) J.D. Corbett, et al.; Biophys. J. 66, 25 (1994) | (5) T.P. Wu, et al.; Nucleic Acids Res. 24, 3472 (1996) | (6) X. Gao, et al.; Bioorg. Chem. 25, 163 (1997) | (7) N. Tamilarasu, et al.; Bioconjug. Chem. 12, 135 (2001) | (8) A.R. Chauduri, et al.; Mech. Ageing Developm. 127, 849 (2006) | (9) Y. Zhang, et al.; Carbohydr. Res. 346, 2156 (2011) | (10) Y. Zhang, et al.; Glycoconj. J. 29, 445 (2012) | (11) Y. Zhang, et al.; Eurp. Food. Res. Technol. 239, 867 (2014) | (12) M. Have, et al.; Plant Biol. 17, 973 (2015) | (13) C. Qiu, et al.; Methods Mol. Biol. 1297, 113 (2015) | (14) R.W. Sabnis; Handbook of Fluorescent Dyes and Probes p224 (2015)
Oral absorption characteristics and mechanisms of a pectin-type polysaccharide from Smilax china L. across the intestinal epithelium: Y. Zhang, et al.; Carbohydr. Polym. 270, 118383 (2021)